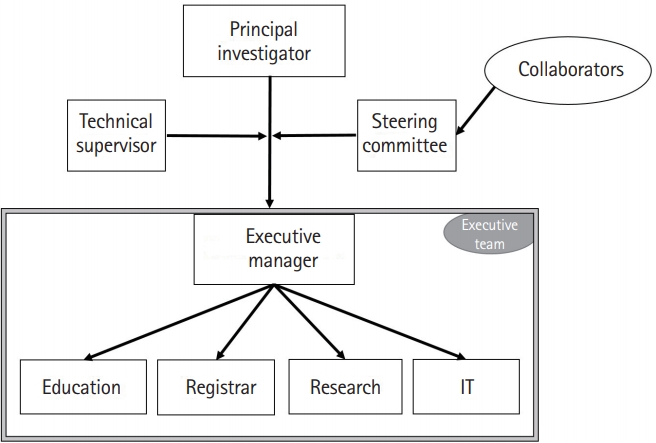
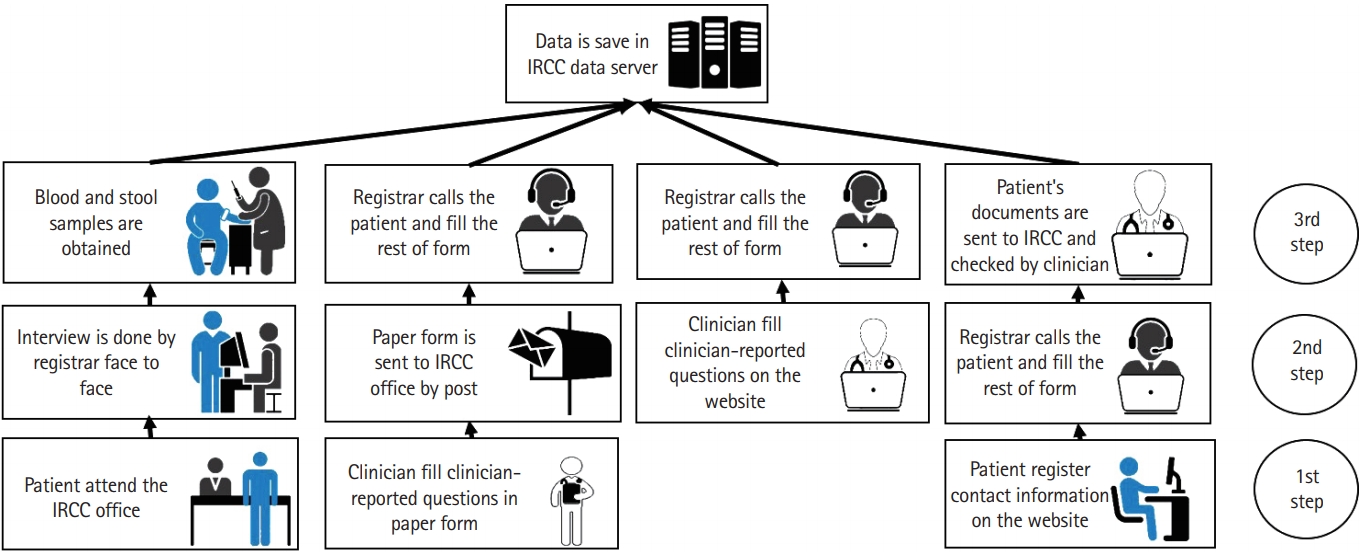
|
 |
- Search
| Intest Res > Volume 17(3); 2019 > Article |
|
Abstract
Background/Aims
Methods
Results
Conclusions
NOTES
FINANCIAL SUPPORT
This work was supported by Deputy of Research of the Ministry of Health and Medical Education, and Iranian Association of Gastroenterology and Hepatology.
AUTHOR CONTRIBUTION
Study design, data analysis, writing the first draft, finalizing the manuscript: Malekzadeh MM. Study design and conduct, data acquisition, revising the manuscript and confirm the final version of manuscript: Sima A, Vahedi H. Study design, revising the manuscript and confirm the final version of manuscript: Alatab S, Zendedel K. Study design, data acquisition, revising the manuscript and confirm the final version of manuscript: Sadeghi A, Malekzadeh R. Data acquisition, revising the manuscript and confirm the final version of manuscript: Daryani NE, Adibi P, Maleki I, Vossoughinia H, Fakheri H, Yazdanbod A, Taghavi SA, Aghazadeh R, Somi MH. Approval of final manuscript: all authors.
ACKNOWLEDGEMENTS
SUPPLEMENTARY MATERIAL
Fig.┬Ā2.
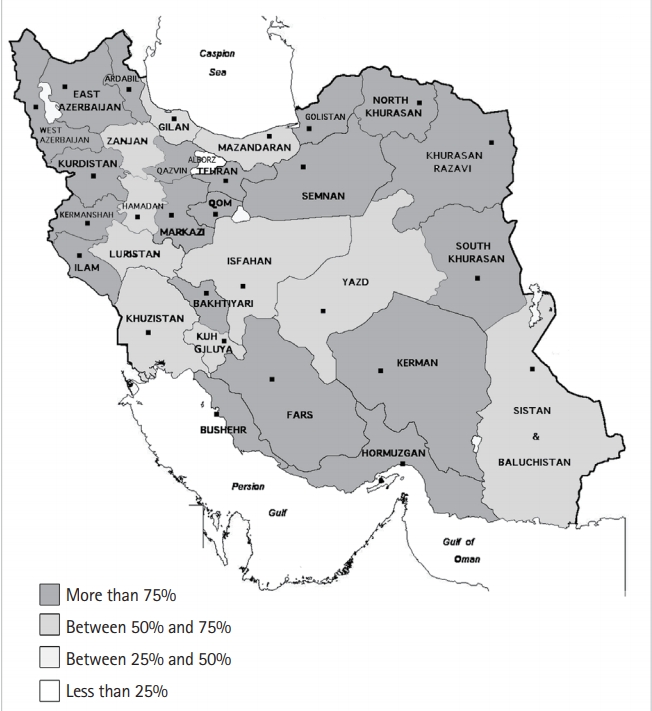
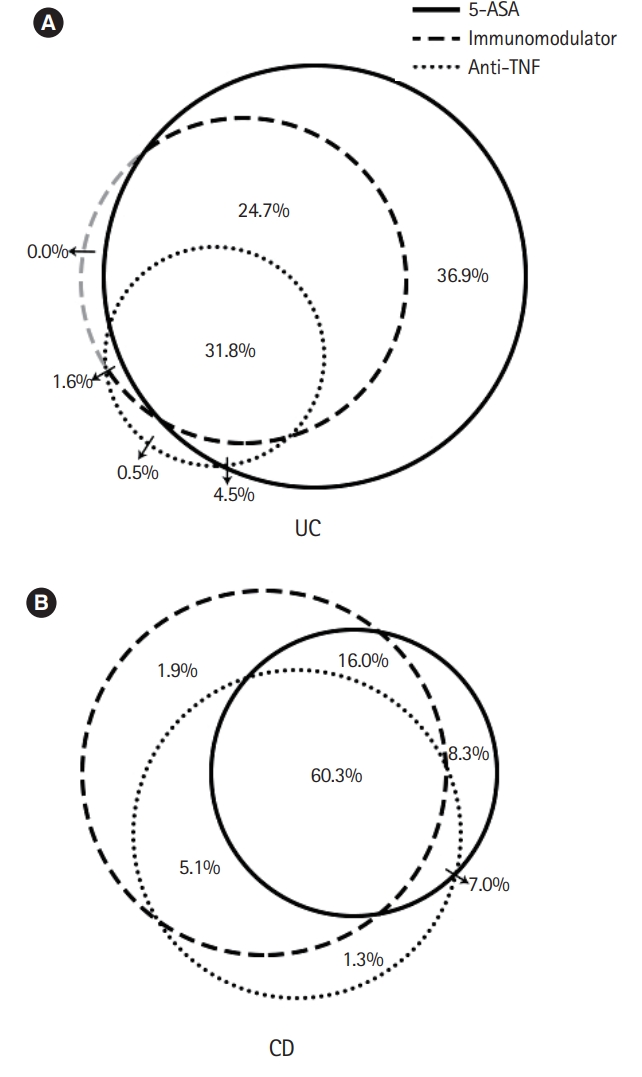
Fig.┬Ā4.
Table┬Ā1.
Table┬Ā2.
| Characteristic | Value |
|---|---|
| Sex | |
| ŌĆāMale | 312 (56.42) |
| ŌĆāFemale | 241 (43.58) |
| Age group (yr) | |
| ŌĆā0-19 | 21 (3.80) |
| ŌĆā20-29 | 106 (19.17) |
| ŌĆā30-39 | 206 (37.25) |
| ŌĆā40-49 | 120 (21.70) |
| ŌĆā50-59 | 61 (11.03) |
| ŌĆā60-69 | 27 (4.88) |
| ŌĆāŌēź70 | 12 (2.17) |
| Education | |
| ŌĆāIlliterate | 6 (1.08) |
| ŌĆāPrimary school | 20 (3.62) |
| ŌĆāMiddle school | 58 (10.49) |
| ŌĆāHigh school | 166 (30.02) |
| ŌĆāAssociate degree | 49 (8.86) |
| ŌĆāBachelor | 170 (30.74) |
| ŌĆāMaster | 65 (11.75) |
| ŌĆāDoctoral | 19 (3.44) |
| Depression | |
| ŌĆāNo | 500 (90.42) |
| ŌĆāYes | 53 (9.58) |
| Appendectomy | |
| ŌĆāNo | 495 (89.51) |
| ŌĆāYes | 58 (10.49) |
| Smoking | |
| ŌĆāNever | 485 (87.7) |
| ŌĆāCurrent user | 36 (6.51) |
| ŌĆāPast user | 32 (5.79) |
| Age (yr) | |
| ŌĆāUC | 39.51┬▒13.25 |
| ŌĆāCD | 36.10┬▒11.78 |
| ŌĆāBoth | 38.58┬▒12.87 |
| Hookah | |
| ŌĆāNever | 509 (92.04) |
| ŌĆāCurrent user | 32 (5.79) |
| ŌĆāPast user | 12 (2.17) |
| Opium | |
| ŌĆāNever | 540 (97.65) |
| ŌĆāCurrent user | 9 (1.63) |
| ŌĆāPast user | 4 (0.72) |
| Ethnicity | |
| ŌĆāFars | 358 (64.74) |
| ŌĆāTurk | 90 (16.27) |
| ŌĆāLor | 33 (5.97) |
| ŌĆāKurd | 35 (6.33) |
| ŌĆāArab | 7 (1.27) |
| ŌĆāOther | 30 (5.42) |
| Family history (1st degree) | |
| ŌĆāUC | 50 (9.04) |
| ŌĆāCD | 15 (2.71) |
| ŌĆāBoth | 65 (11.75) |
| Family history (2nd degree) | |
| ŌĆāUC | 29 (5.24) |
| ŌĆāCD | 9 (1.62) |
| ŌĆāBoth | 38 (6.87) |
| Disease activity during 2 weeks before enrollmenta | |
| ŌĆāQuiescent | 289 (52.26) |
| ŌĆāActive | 264 (47.74) |
| Disease activity during 6 months before enrollmentb | |
| ŌĆāQuiescent | 341 (61.66) |
| ŌĆāActive | 212 (38.34) |
| Age at diagnosis (yr) | |
| ŌĆāUC | 31.36┬▒12.16 |
| ŌĆāCD | 27.92┬▒11.32 |
| ŌĆāBoth | 30.43┬▒11.98 |