 |
 |
- Search
| Intest Res > Volume 19(1); 2021 > Article |
|
Abstract
Background/Aims
Methods
Results
ADDITIONAL INFORMATION
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contribution
Conceptualization: Makharia GK, Agarwal A. Methodology: Agarwal A, Makharia GK, Singh A, Mehtab W. Formal analysis: Agarwal A, Makharia GK, Chauhan A. Project administration: Makharia GK. Visualization: Makharia GK, Agarwal A. Writing - original draft: Agarwal A, Singh A, Chauhan A, Rajput MS. Writing - review and editing: Agarwal A, Makharia GK, Ahuja V, Makharia GK, Singh N, Mehtab W, Gupta V. Approval of final manuscript: all authors.
Fig. 1.
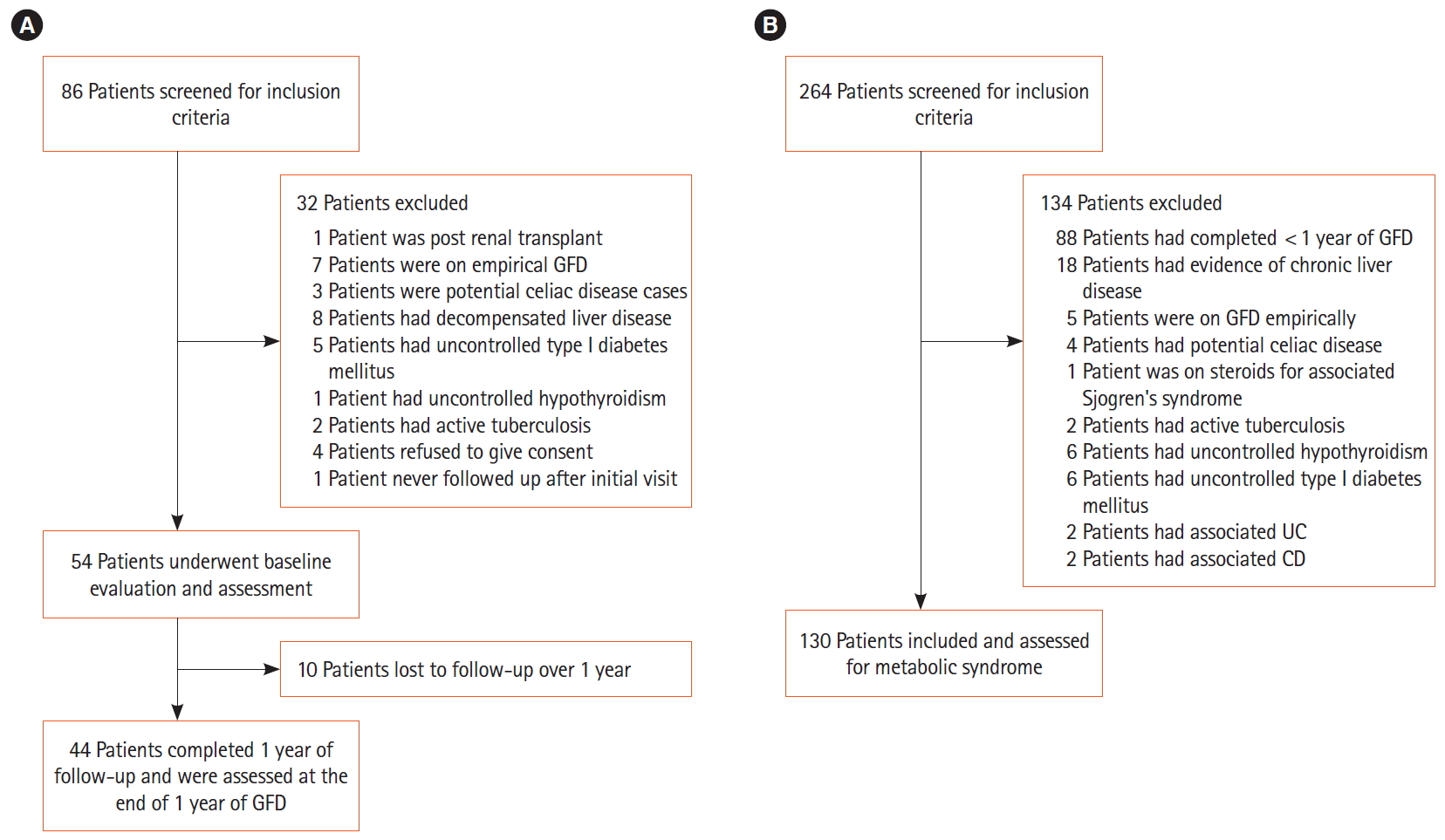
Fig. 2.
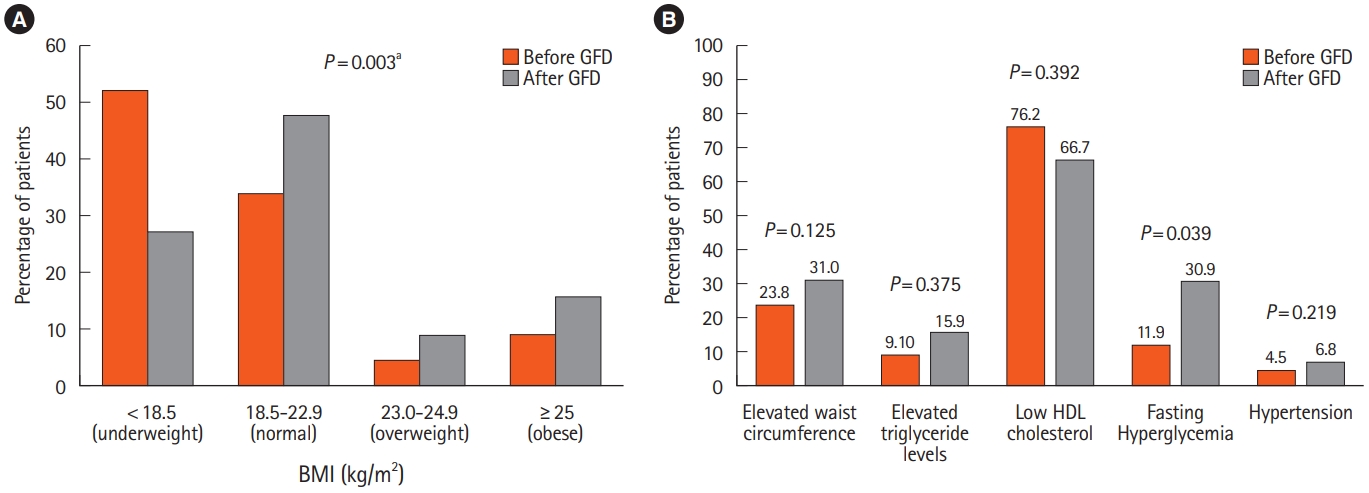
Table 1.
| Characteristic | Before GFD (n = 44) | After GFD (n = 44) | P-valuea | Patients on long-term GFD (n = 130) |
|---|---|---|---|---|
| Sex | - | |||
| Male | 18 (41.0) | 18 (41.0) | 53 (40.8) | |
| Female | 26 (59.0) | 26 (59.0) | 77 (59.2) | |
| Age (yr) | 29.5 ± 11.3 | - | - | 33.0 ± 13.1 |
| Hemoglobin (g/dL) | 10.4 ± 2.6 | 12.4 ± 1.8 | < 0.001 | 12.1 ± 1.8 |
| Hemoglobin categories (g/dL) | < 0.001 | |||
| <8 | 10 (23.3) | 0 | 2 (1.6) | |
| 8-12 | 17 (39.5) | 19 (43.2) | 59 (45.4) | |
| > 12 | 16 (37.2) | 25 (56.8) | 67 (51.5) | |
| Corpuscular volume (fL/cell) | 77.2 ± 10.6 | 81.0 ± 7.0 | 0.009 | 83.4 ± 8.8 |
| Corpuscular hemoglobin (pg/cell) | 25.2 ± 4.3 | 28.2 ± 10.3 | 0.080 | 27.2 ± 2.9 |
| Corpuscular hemoglobin concentration (g/dL) | 30.8 ± 1.7 | 31.4 ± 2.1 | 0.014 | 31.5 ± 1.8 |
| AST (U/L) | 32.8 ± 16.0 | 31.7 ± 14.8 | 0.420 | 28.5 (11.0-83.0) |
| ALT (U/L) | 30.9 ± 13.8 | 30.9 ± 19.1 | 0.974 | 28.5 (8.0-73.0) |
| Transaminitis (more than 1.5 times elevated) | 4/42 (9.5) | 3/44 (6.8) | 0.240 | 10/130 (7.7) |
| IgAtTG elevation (folds rise) | 9.2 (1.5-40.5) | 2.1 (0.1-14.2) | < 0.001 | 0.9 (0.1-55.3) |
| IgAtTG more than one folds rise | - | 30/44 (68.2) | - | 56/125 (44.8) |
| Compliance with GFD | - | - | ||
| Excellent | 23 (52.3) | 90 (69.8) | ||
| Good | 14 (31.8) | 27 (20.9) | ||
| Poor | 1 (2.3) | 9 (6.9) | ||
| Very poor | 6 (11.1) | 3 (2.3) | ||
| Height (cm) | 158.7 ± 11.0 | 159.0 ± 10.7 | 0.300 | 156.7 ± 21.7 |
| Weight (kg) | 49.0 ± 13.7 | 53.2 ± 13.3 | < 0.001 | 57.1 ± 12.8 |
| BMI (kg/m2) | 19.3 ± 4.2 | 20.9 ± 4.1 | < 0.001 | 22.5 ± 4.3 |
| BMI categories (kg/m2) | 0.003 | |||
| < 18.5 | 23 (52.3) | 12 (27.3) | 25 (19.2) | |
| 18.5-22.9 | 15 (34.1) | 21 (47.7) | 54 (41.5) | |
| 23.0-24.9 | 2 (4.54) | 4 (9.1) | 18 (13.8) | |
| > 25.0 | 4 (9.1) | 7 (15.9) | 33 (25.4) | |
| Waist circumference (cm) | 76.9 ± 10.9 | 80.1 ± 12.5 | 0.003 | 82.1 ± 10.6 |
| Elevated waist circumference | 0.125 | |||
| Male > 90 cm | 3 (16.7) | 3 (16.7) | 11 (20.8) | |
| Female > 80 cm | 7 (26.9) | 11 (42.3) | 44 (57.1) | |
| Hip circumference (cm) | 84.6 ± 7.9 | 88.0 ± 8.7 | < 0.001 | 90.4 ± 9.2 |
| Body fat % | 10.0 (2.5-31.0) | 12.0 (1.8-37.0) | 0.001 | 18.0 (1.1-40.0) |
| Elevated body fat percentage | 0.713 | |||
| Male > 25% | 2 (11.1) | 3 (16.7) | 6 (11.3) | |
| Female > 30% | 1 (3.8) | 2 (7.7) | 19 (24.7) | |
| Fat mass index (kg/m2) | 1.8 (0.4-9.1) | 2.4 (0.3-10.5) | 0.003 | 3.8 (0.2-18.7) |
| Elevated fat mass index (kg/m2) | 0.713 | |||
| Male >7 | 2 (11.1) | 3 (16.7) | 6 (11.3) | |
| Female > 7.9 | 1 (3.8) | 2 (7.7) | 19 (24.7) | |
| Fasting sugar level (mg/dL) | 91.8 ± 8.5 | 93.5 ± 9.1 | 0.226 | 94.9 ± 10.7 |
| Fasting sugar level > 100 mg/dL | 5/42 (11.9) | 13/42 (31.0) | 0.039 | 41/119 (34.5) |
| Total cholesterol (mg/dL) | 145.4 ± 38.7 | 158.0 ± 35.8 | 0.002 | 166.7 ± 39.0 |
| HDL (mg/dL) | 39.5 ± 10.3 | 42.8 ± 8.7 | 0.058 | 44.8 ± 9.8 |
| Low HDL | 0.392 | |||
| Male (< 40 mg/dL) | 10 (55.6) | 6 (33.3) | 15 (28.3) | |
| Female (< 50 mg/dL) | 22/25 (88.0) | 22 (84.6) | 46/74 (62.1) | |
| LDL (mg/dL) | 88.6 ± 28.4 | 97.0 ± 24.0 | 0.001 | 108.7 ± 33.4 |
| VLDL (mg/dL) | 13 (6-34) | 15 (10-32) | 0.159 | 14 (7-42) |
| Triglyceride (mg/dL) | 83 (38-360) | 97 (53-436) | 0.014 | 105.4 ± 48.9 |
| Triglyceride > 150 mg/dL | 4 (9.1) | 7 (15.9) | 0.375 | 16/121 (13.2) |
| Hypertension | 2 (4.5) | 3 (6.8) | 1.000 | 6/130 (4.6) |
| LSM (kPa) | 5.1 ± 1.9 | 4.8 ± 1.8 | 0.098 | 4.9 ± 1.2 |
| CAP (dB) | 213.5 ± 46.9 | 230.8 ± 50.1 | 0.001 | 226.7 ± 50.2 |
| CAP categories (dB) | 0.002 | |||
| < 263 (no NAFLD) | 36/42 (85.7) | 31 (70.5) | 100 (76.9) | |
| ≥ 263 (NAFLD) | 6/42 (14.3) | 13 (29.5) | 30 (23.1) | |
| 263.0-286.9 (grade I) | 3/42 (7.1) | 5 (11.4) | 15 (11.5) | |
| 287.0-295.5 (grade II) | 2/42 (4.8) | 0 | 5 (3.8) | |
| ≥ 296.0 (grade III) | 1/42 (2.4) | 8 (18.2) | 10 (7.7) |
GFD, gluten-free diet; AST, aspartate aminotransferase; ALT, alanine aminotransferase; IgAtTG, IgA anti-tissue transglutaminase antibody; BMI, body mass index; HDL, high density lipoprotein; LDL, low density lipoprotein; VLDL, very low-density lipoprotein; LSM, liver stiffness measurement; CAP, controlled attenuation parameter; NAFLD, nonalcoholic fatty liver disease; SD, standard deviation.
Table 2.
| Asian Indian Consensus criteria |
Group 1 |
Group 2 |
||
|---|---|---|---|---|
| Before GFD | After 1 year of GFD | P-valuea | Patients on long-term GFD | |
| Abdominal obesity, assessed as waist circumference | 0.125 | |||
| Male > 90 cm | 3/18 (16.7) | 3/18 (16.7) | 11/49 (22.4) | |
| Female > 80 cm | 7/26 (26.9) | 11/26 (42.3) | 40/65 (61.5) | |
| Triglycerides > 150 mg/dL | 4/44 (9.1) | 7/44 (15.9) | 0.375 | 16/114 (14.0) |
| HDL cholesterol | 0.392 | |||
| Male < 40 mg/dL | 10/18 (55.6) | 6/18 (33.3) | 15/49 (30.6) | |
| Female < 50 mg/dL | 22/25 (88.0) | 22/26 (84.6) | 43/65 (66.2) | |
| Blood pressure > 130/85 mmHg | 2/44 (4.5) | 3/44 (6.8) | 1.000 | 6/114 (5.3) |
| Fasting blood sugar ≥ 100 mg/dL | 5/44 (11.9) | 13/44 (30.9) | 0.039 | 40/114 (35.1) |
| ≥ 3 Criteria satisfied | 5/44 (11.4) | 9/44 (18.2) | 0.219 | 30/114 (26.3) |








