The role of microbiome in colorectal carcinogenesis and its clinical potential as a target for cancer treatment
Article information
Abstract
The role of gut microbiome-intestinal immune complex in the development of colorectal cancer and its progression is well recognized. Accordingly, certain microbial strains tend to colonize or vanish in patients with colorectal cancer. Probiotics, prebiotics, and synbiotics are expected to exhibit both anti-tumor effects and chemopreventive effects during cancer treatment through mechanisms such as xenometabolism, immune interactions, and altered eco-community. Microbial modulation can also be safely used to prevent complications during peri-operational periods of colorectal surgery. A deeper understanding of the role of intestinal microbiota as a target for colorectal cancer treatment will lead the way to a better prognosis for colorectal cancer patients.
INTRODUCTION
Colorectal cancer (CRC) is the second most common cancer in men and the third in women globally [1]. It is also the second most commonly diagnosed cancer after gastric cancer in South Korea, with 30,000 new diagnoses and 8,700 deaths each year [2].
There are many long-term and late effects during the treatment of CRC, including chronic peripheral neuropathy, secondary cancers, bowel dysfunction such as nausea and diarrhea, and psychological issues such as depression and anxiety [3]. Most current guidelines focus on diagnosis and treatment of the tumor itself, but there is also a need for a “better adjunctive care” during the colon cancer treatment, and one of them is the “modulation of gut microbiota.”
The human intestine is an organ inhabited by billions of microorganisms, of which 10 [14] are in the colon [4,5]. These microorganisms are collectively referred to as “microbiota.” Their genes are called “microbiome” or “the 2nd genome” of humans. The importance of the gut microbiome in colorectal carcinogenesis is relatively well known. Active research is underway on the effects of microbial modulation through pre- and probiotics during cancer treatment.
This review is aimed to attract attention regarding the beneficial effects of intestinal microbial modulation during the treatment course of CRC. A deeper understanding of this topic will help the treatment of CRC patients. In the long term, it will help us develop tailored therapy according to characteristics of patient-specific intestinal microbiota and their immunologic states.
GUT MICROBIOME-INTESTINAL IMMUNE COMPLEX
“All disease begins in the gut.” As mentioned in the 3rd century BC by Hippocrates, the gut microbiome plays surprisingly diverse roles. They contribute to the body’s energy metabolism, synthesis of vitamins and other essential nutrients, signaling the endocrine system, preventing colonization of harmful bacteria, regulating the immune system, and contributing to the metabolism of xenobiotic compounds [6,7].
Maintaining the intestinal mucosa’s immunological homeostasis begins with a challenging task of discriminating rare pathogenic species from billions of harmless microbes. During this process, both innate and adaptive immune responses prevent colonization of pathogens and induce local and systemic inflammatory responses to foreign microbial and dietary antigens. Gut-associated lymphoid tissue is the backbone of immune surveillance and defense mechanism. It consists of Peyer’s patch, appendix, isolated lymphoid follicle, and mesenteric lymph nodes. It is responsible for both activation and suppression of the mucosal immune system. The presence of intestinal bacteria is also essential for the development and maturation of the gut-associated lymphoid tissue [8].
When pathogens or symbiotic bacteria cross the epithelial barrier and enter the host, they first encounter macrophages. Antigens that are not phagocytosed by macrophages are captured by intestinal dendritic cells and migrated from the intestine to mesenteric lymph nodes, triggering a differentiation process that leads to the production of regulatory T cells (Treg), T-helper 17 (Th17), and IgA secreting B-cells [9]. According to a study using germ-free mice, the intestinal microbial community plays an important role in forming adequate mucosal immunity. Compared to specific pathogen-free mice, germ-free mice had fewer intraepithelial lymphocytes, less sIgA secretion in the lamina propria, and fewer Treg production [10].
Foxp3+ Treg cells play a key role in intestinal immune tolerance mechanisms [11]. CD4+Foxp3+ Treg cells derived from naive CD4+ T cells in both the thymus (nTreg) and intestine (iTreg) can help maintain immune unresponsiveness to autoantigens and suppress excessive immune reactions that might be harmful to the host. In an environment where Treg cells are abnormally present, proper immune tolerance is not induced, resulting in hypersensitive reactions [12,13]. Bacteroides fragilis, a type of symbiotic bacteria, can produce polysaccharide A, inhibit interleukin17 (IL-17) production from Th17 cells, and enhance the activity of iTregs, leading to anti-inflammatory effects. polysaccharide A can also induce the transformation of CD4+ T cells into Foxp3+ Tregs. It can also down-regulate the production of pro-inflammatory Th17 cells [14,15]. B. fragilis is known to be able to improve colitis in a mouse model. However, Bacteroides spp. are thought to have ambivalence as B. fragilis toxin (BFT) can cause inflammatory bowel disease by altering the function of epithelial tight junctions [16,17].
Th17 cells in the mucous membrane of the small intestine are vital in protecting the mucosal surface from microbial pathogens. However, they are also notorious for inducing autoimmune inflammation when they are activated by IL-23 [18,19]. Th17 cells seem to have an opposite function of Tregs [20-23]. Recently, it has been reported that Th17 cells are a unique CD4+ T-helper subset characterized by the production of IL-17, which can promote inflammation against a variety of pathogens. Specific intestinal microflora that can induce small intestinal Th17 cells are known as “segmented filamentous bacteria.” [24,25]
Another role of intestinal microbes is that they can ferment polysaccharides (such as resistant starch, oligosaccharides, inulin, etc.) that humans cannot normally digest or absorb, resulting in the production of short-chain fatty acids (SCFA) [26]. These SCFAs also contribute to the activation of several types of immune cells and play an important role in the differentiation of Treg cells [27,28].
As described above, it has been suggested that modulating the gut microbiome may prevent or worsen various types of inflammatory and allergic diseases because it can alter the differentiation of immunologic cells and modulate the production of SCFA by promoting colonization of beneficial bacteria [29,30].
MICROBIAL SIGNATURES OF CRC
The gut microbiota has a large diversity of microbial populations composed of bacteria, archaea, eukaryotes, and viruses. Both tissue and fecal samples provide information on the structure of bacterial populations. Analyzing tissue samples will show a more direct relationship between colon cancer’s pathophysiology and the gut microbiota. However, since sampling the intestinal mucosa is invasive, research using biopsy samples from normal mucosa is particularly limited. Therefore, most studies rely on fecal samples to analyze the distribution and diversity of intestinal microorganisms.
Most bacteria that reside in the gut are strictly anaerobic, and therefore they cannot be grown or cultured. The ability to identify bacteria using culture-independent methods was a huge advance in the microbiome field. Sequencing V1–V3 or V3–V5 variable regions of bacterial 16S ribosomal RNA has become a standard method for identifying bacterial populations or operational taxonomic units known to generally represent bacterial species whose sequences share more than 97% identities to each other [31]. Using the 16s rRNA target gene sequencing method and the recently introduced shotgun metagenomic sequencing, we can obtain more detailed information on the intestinal microbiome.
The gut microbiota not only promotes intestinal homeostasis and anti-tumor responses, but also contributes to genotoxic effects that can lead to carcinogenesis by causing chronic dysregulated inflammation [32]. Whether the gut microbiota will form a healthy symbiosis relationship with the host or promote colon cancer ultimately depends on the composition of gut microbiota and the balance between harmful bacterial populations within the microbiome. However, whether this “dysbiosis” might precede or cause CRC remains unclear. Studies on mice strongly suggest that gut microbiota may modulate susceptibility to CRC and may serve as both early diagnostic biomarkers and therapeutic targets. Such clinical research should be done considering the influence of each individual’s race, lifestyle, diet, sample type, location of the tissue sampled, and gut microbial ecosystem. Several notable shifts in the phylum level have been reported in the intestinal bacterial community of CRC patients. Among them, Bacteroides, Fusobacteria, and Proteobacteria are increased while Firmicutes are decreased in both intestinal mucosa and feces of the colon cancer patients [33,34]. More specifically, enterotoxic strains of B. fragilis and polyketide synthase (PKS) positive strains of Escherichia coli, Fusobacterium nucleatum are 3 most well-known strains in colorectal tumorigenesis (Fig. 1) [35,36].

Step-by-step mechanisms of microbiota inducing colorectal cancer. Proposed mechanisms by which commensal gut microbiota interact with gastrointestinal epithelium and induce colorectal cancer. (A) Fusobacterium nucleatum. (B) Enterotoxigenic Bacteroides fragilis. (C) PKS + Escherichia coli. LPS, lipopolysaccharide; TLR4, Toll-like receptor 4; TIGIT, T-cell immunoglobulin and ITIM domain; CEC, colonic epithelial cell; NK cell, natural killer cell; IL, interleukin; STAT3, signal transducer and activator of transcription 3; CXCL, chemokine (C-X-C motif) ligand; CXCL1, chemokine (C-X-C motif) ligand 1 peptide; APC, adenomatosis polyposis coli; PKS, polyketide synthase.
Colonization of enterotoxin positive BFT (ETBF) has long been studied to be associated with diarrhea and gastrointestinal inflammation. ETBF also induced early carcinogenesis in mice models. ETBF coats tumors and recruits other bacteria to form a biofilm. Recently, increased colonization ETBF biofilms coating early human CRCs was confirmed [37]. BFT induces Th-17 mediated colitis and IL17-dependent carcinogenic inflammation through an accumulation of Treg cells.
E. coli species can be divided into 4 phylotypes (A, B1, B2, and D). Bonnet et al. [38] and Raisch et al. [39] confirmed that mucous membranes of cancer patients were much more abundant in E. coli subgroup B2. When B2 phylotype E. coli are incubated in vitro with various epithelial cell lines, they can arrest the epithelial cell cycle and force them to enter senescence [40-42]. Such effect is due to a group of compounds collectively named cyclomodulins that can introduce double-strand DNA breaks in target cells. These cyclomodulins include cytolethal distending toxin, cytotoxic necrotizing factor, and the best known “colibactin” produced by the PKS locus [40,43,44]. Colibactin is most likely a combination of hybrid molecules containing both a peptide and a polyketide produced in the gut by PKS positive E. coli [45]. Transient infection of cultured epithelial cells with PKS positive E. coli can induce chromosomal aberrations and increase mutation frequency rates. Correspondingly, in animal models of carcinogenesis, exposure to PKS can induce DNA strand breaks and lead to tumor generation [36,46].
While many independent studies have identified specific operational taxonomic units that can differentiate between healthy and CRC patients, F. nucleatum related to periodontal disease have gained attention due to their association with CRC [47,48]. Its prevalence has been reported to be gradually increasing as the disease progresses from colon polyp to CRC [48]. Studies in mice have shown that F. nucleatum can directly promote tumor growth [49]. F. nucleatum can reduce natural killer cell-mediated tumor killing by interacting with receptor TIGIT (T-cell immunoglobulin and ITIM domain) and inhibiting anti-tumorous natural killer cell activity [50]. In addition, E-cadherin-mediated interactions with CRC epithelial cells can induce cell proliferation via Wnt signaling and avoid immune surveillance [51]. Interestingly, there are reports that F. nucleatum is specifically related to microsatellite instability high tumors and that colonization of F. nucleatum is associated with relatively shorter cancer survival [51], making it suitable for use as a biomarker. Indeed, CRC with high loads of F. nucleatum, cells are more resistant to oxaliplatin by activating autophagy through Toll-like receptor 4 expressed on CRC cells [52]. Fap2 and FadA of F. nucleatum are proteins facilitate the binding to TIGIT and E-cadherin and enriches tumor proliferation. They could be used as potential targets for treating and detecting early CRC. However, since F. nucleatum is not universally present in all CRCs and its DNA is only detected in about 13% of all CRC cases, the use of F. nucleatum-derived proteins as a treatment target should be individualized.
Questions remain on how and when these tumor-related microbial act in the carcinogenesis of CRC species (sequentially, tandemly, or simultaneously). In addition, potential species such as Streptococcus gallolyticus and Enterococcus faecalis should be investigated. We must emphasize that further research to understand how the microbial species’ arrivals and departures in intestinal mucosa affect the tumor and its progression as time passes through is desperately needed beyond simply listing which species coexist with CRC.
BENEFICIAL EFFECTS OF PRE/PRO/SYNBIOTICS IN THE TREATMENT OF CRC
1. Probiotics
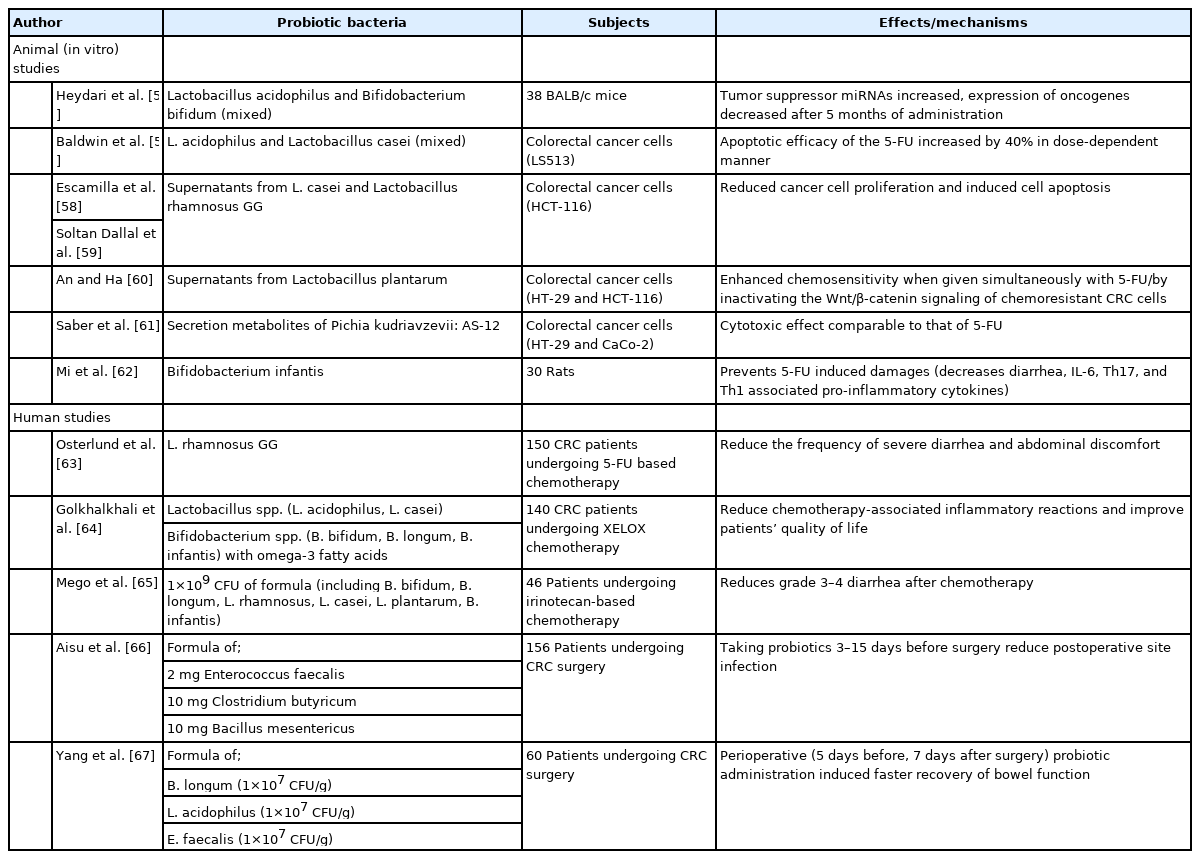
According to the Food and Agriculture Organization of the United Nations and the World Health Organization, probiotics are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host.” [53] Most studies before 2010 focused on the role of probiotics and prebiotics in the prevention of colon cancer. However, recent studies have introduced ways to utilize them even during treatment (Table 1). Probiotics are expected to exert anti-tumor and anti-mutagenic activities during cancer treatment since gut microbiota seems to be implicated in chemotherapy efficacy through various mechanisms, including xenometabolism, immune interactions, and altered community structure [54].
Using an animal colon cancer model, Heydari et al. [55] have reported that levels of tumor suppressor miRNAs such as miR-26b, miR-18a, APC, and PTEN are increased after 5 months of administration of probiotics (Lactobacillus acidophilus, Bifidobacterium bifidum). At the same time, expression levels of oncogenes such as miR135b and KRAS are decreased. Baldwin et al. [56] have evaluated the difference in apoptotic activity of 5-fluorouracil (5-FU) in CRC cell lines using live or inactive lactic acid bacteria (LAB) such as L. acidophilus and Lactobacillus casei at different concentrations. As a result, 5-FU efficacy was observed to increase up to 40% in a dose-dependent manner with LAB. This was thought to be due to the ability of LAB to convert lactate and acetate to butyrate, a well-known anticarcinogenic agent [57].
Meanwhile, a study has found that secretory metabolites of LAB also have inhibitory effects on the invasiveness of the human colorectal cell line (HCT-116) [58]. That study was done using extracts of cell-free supernatants of L. casei and Lactobacillus rhamnosus GG (LGG). Soltan Dallal et al. [59] have compared direct effects on colorectal tumor cells between supernatants of Lactobacillus species and bacterial extracts in terms of proliferation, necrosis, apoptosis, migration, and invasion respectively. As a result, lactobacilli supernatants reduced cancer cell proliferation and induced cell apoptosis without inducing cell necrosis, while lactobacilli extract induced cell necrosis. However, both acted positively on cancer cell migration and invasion.
In an in vitro study conducted by An and Ha [60] with 5-FU resistant colon cancer cell lines (HT-29 and HCT-116), L. plantarum cell-free supernatants enhanced the chemosensitivity when given simultaneously with 5-FU. In addition, various types of Lactobacillus strains and their supernatants were tested against human colon cancer cell lines. Among them, BCRC17010 strain showed the most promising adhesion ability, longer survival in the gastrointestinal tract, and increased lactate dehydrogenase release [68]. There might be differences in their abilities to act as an adjunctive agent depending on the substrain of Lactobacillus. On the other hand, one in vitro study has indicated that the direct cytotoxic ability of bacterial secretion metabolites could be comparable to that of 5-FU. Saber et al. [61] have reported that methanolic extract of secreted metabolites of Pichia kudriavzevii AS-12 (MEPK) can increase the expression of pro-apoptotic mediates in HT-29 and CaCo-2 cell lines, suggesting that it has potential as an anticancer agent.
Chemoprotective effect and anti-CRC properties of Bifidobacterium infantis have also been evaluated in a mouse model [62]. Similar to results of previous studies, chemotherapy-induced health damages were hindered by a strain of probiotics with changes in T-cell immunity profiles such as decreased IL-6, Th17, and Th1 cell-associated cytokines and increased Foxp3+, Tregs, and so on.
There is a safety issue on the use of probiotics in CRC patients. Probiotics are generally well tolerated in healthy subjects, but in patients with damaged intestinal barrier or compromised immunity, such protection may fail and lead to bacterial translocation, systemic infection and antimicrobial resistance. In a systematic review and meta-analysis, the study suggests probiotics use may be beneficial but 5 case reports showed probiotic-related bacteraemia, fungemia. Despite these case reports, current evidence does not suggest an absolute contraindication on probiotic in cancer patients.
2. Next-Generation Probiotics
Next-generation probiotics (NGPs) are defined as “live microorganisms identified on the basis of comparative microbiota analysis that, when administered in adequate amounts, confer a health benefit on the host.” Apart from traditional probiotics that have long been isolated from fermented foods, NGPs have been recently isolated due to the development of tools that can now identify and modify these commensal bacteria. NGPs such as Bifidobacterium spp., B. fragilis, Akkermansia municiphila, and Faecalibacterium prausnitzii are opening novel therapeutic horizons in CRC treatment [69].
Some strains of Bifidobacterium species may enhance the efficacy of cancer therapy with immune checkpoint inhibitors (ICI). Especially, B. fragilis may increase the efficacy of ICI therapy [70]. However, be cautious when using B. fragilis as enterotoxin-containing B. fragilis has been associated with a negative result, leading to paradoxical CRC development. It has been shown that A. municiphila, contributes to a better efficiency of PD-1 based immunotherapy in mouse models [71,72]. F. prausnitzii is the one which takes part in butyrate production and may bring beneficial effects on CRC treatment. However, these ICI-related probiotic studies have limitations in that they have not been conducted in CRC cell lines. With the introduction of NGPs, probiotics are expected to have an endless potential in the treatment of CRC.
3. Probiotics Can Improve Chemotherapy-Induced Toxicities
The administration of probiotic strains can reduce side effects of anticancer therapy, especially adverse events after chemotherapy and radiotherapy. This hypothesis has been reinforced by many studies [73], showing that microbiome modulation through alimentation or probiotic supplementation could reduce chemotherapy toxicity and other subsequent side effects in mice and humans. Many papers have suggested that microbemediated xenometabolism could be linked to an increase of chemotherapeutic toxicity, leading to a decrease in treatment efficacy [74,75].
Chemotherapy regimens based on 5-FU is frequently associated with the risk of intestinal mucositis and diarrhea. The most serious case of toxicity associated with death was reported in Japan following 5-FU and sorivudine combined therapy. Besides, intestinal mucositis can be induced by other various chemotherapy agents (Irinotecan, Doxorubicin, etc.). The accumulation of active xenometabolite (SN-38) from Bacteroides and other β-glucuronidase-producing bacteria is believed to be involved. In a Finnish study [63], 150 CRC patients who received 5-FU based postoperative adjuvant chemotherapy were randomly assigned to receive chemotherapy for 24 weeks with or without LGG supplementation. Patients who received Lactobacillus had less grade (3 or 4) diarrhea and fewer chemotherapy dose reductions. No influence on chemotherapy tolerability was reported. Probiotics can produce several organic acids and SCFAs that can help maintain a low pH in the intestinal lumen, one of key factors in protecting intestinal epithelial cells. In a double-blinded, randomized clinical trial of CRC patients undergoing XELOX (capecitabine plus oxaliplatin) chemotherapy, the Malaysian research team has reported that mixed probiotics (L. casei, L. acidophilus, L. lactis, B. bifidum, etc.) for 8 weeks can significantly reduce chemotherapy-associated inflammatory reactions with IL-6 reduction and improve patients’ quality of life [64]. The overall safety of the use of probiotics for the prevention and treatment of chemotherapy-induced diarrhea has been verified through a systemic review and meta-analysis [76].
The efficacy of probiotics can also be found in chemotherapy-induced neutropenic patients. Most infections in these neutropenic patients are caused by endogenous flora. The main route is through intestinal mucosa. A competitive inhibition of bowel colonization between pathogenic microorganisms and probiotics might be a useful prevention tool for these cancer patients. Therefore, several clinical trials have been performed with the postulation that augmentation of colonization resistance by LAB might be an effective and cost-effective way for prevention of opportunistic infection in leukopenic patients. Probiotics composed of Lactobacillus spp. and Bifidobacterium spp. are generally regarded as safe in neutropenic patients [77]. However, larger randomized clinical studies should be followed.
4. Perioperative Use of Probiotics
Probiotics may also effectively protect the intestinal mucosal barrier in CRC patients undergoing surgical procedures. Many clinical studies studied on the effectiveness of perioperative probiotic supplementation in CRC patients. Taking probiotics from 3 to 15 days before surgery reduced post-operational site infection (Aisu et al. [66]), promoted recovery to normal gut function (Tan et al. [78]), and reduced postoperative diarrhea (Yang et al. [67]). A randomized, double-blind study [79] has reported that probiotics supplementation can significantly reduce the rate of all major postoperative complications of colorectal surgery (probiotics 28.6% vs. placebo 48.8%, P=0.010). A systemic review and meta-analysis [80] also concluded that the administration of probiotics peri-operatively can reduce the infection rate by half and the incidence of pneumonia.
5. Prebiotics
Prebiotics are selectively fermentable, non-digestible oligosaccharides, or ingredients that can cause alterations in the composition and activity of gut microbiota conferring health benefits. Prebiotics are carbohydrates including fructooligosaccharides (FOS), xylooligosaccharides, galactooligosaccharides (GOS), inulin, and fructans. FOS and GOS have been compounded mainly investigated as prebiotics. These compounds possess many beneficial properties, including stimulating beneficial indigenous gut bacteria, leading to the production of SCFAs, regulating immune response, controlling gene expression in bacterial cells, improving absorption of micronutrients in colon, and modulating xenobiotic-metabolizing enzymes in colon (Table 2) [80]. Prebiotic inulin enriched with oligofructose combined with probiotics LGG and Bifidobacterium lactis can exert an antitumorigenic activity in azoxymethane-induced colon carcinogenesis in rats. A clinical study of preoperative use of prebiotics in CRC patients demonstrated that it improved the abundancy of commensal microbiota and improved serum immunologic indicators as well [81]. However, there is currently little clinical research on the effectiveness of prebiotics in colon cancer treatment. Bacteroides is suggested to be a relevant bacterial species for further research on the mechanism of prebiotics. It should be emphasized that prebiotics such as β (1–4) GOS, lactulose, and FOS produced by transglycosylation of β-galactosidases or β-glucosidases are expected to have their role in CRC prevention [82].
6. Synbiotics
Synbiotics refer to food ingredients or dietary supplements combining probiotics and prebiotics in the form of synergism. The administration of synbiotics in CRC seems to be useful probably due to their immunomodulatory properties and their ability to reduce rates of postoperative infections. The administration of a cocktail consisting of oligofructose-maltodextrin (prebiotics) enriched L. acidophilus, B. bifidum, and Bifidobacteria infantum to rats decreased cancer growth, increased mucin secretion, preservation of tight junctions, and inhibition of inflammation [83]. This cocktail also modulated gut microbiota compositions. Using a recent colon-specific cancer mouse model, it has been found that treatment with synbiotics can suppress dextran sodium sulfate-induced colitis in CDX2P-Cre; APC+/flox mice, thereby reducing mortality and inhibiting tumorigenesis [84]. In a clinical study of 37 CRC patients, administering a synbiotic formula (LGG, B. lactis Bb12, inulin) can decrease tumor proliferation and lead to a positive change of the composition of intestinal microbiota [85].
CONCLUSION
The role of microbiome in colorectal carcinogenesis is evident and its potential as a treatment target is also promising. In summary, probiotic strains such as B. infantis, LGG, L. acidophilus and L. casei are expected to play an adjunctive role in the future treatment of CRC through cancer cell immunomodulation and chemoprotective effects. However, before it can be acknowledged as an established anticancer therapy, the following points should be elucidated before modulating intestinal ecosystem in CRC patients: types of probiotic/prebiotic strains we can choose, their optimal concentrations, duration of therapy, and supplementation method (by dietary habit change, oral pills of pro-, pre-, synbiotics) to bring out the best clinical outcomes. In addition, prospective clinical studies revealing how the gut flora and colon cancer interact over time are needed to explore the exact mechanism for their effectiveness.
Notes
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Author Contribution
Data curation, original draft: Kim SH. Conceptualization, review and editing: Lim YJ. Approval of final manuscript: all authors.