 |
 |
- Search
| Intest Res > Volume 19(4); 2021 > Article |
|
Abstract
Immune checkpoint inhibitor therapies such as ipilimumab, are increasingly being used as a treatment option for a variety of cancers, including metastatic melanoma and have demonstrated effectively a prolonged survival. These agents have an immunological mode of action that predisposes patients to a number of immune-related adverse events, colitis being one of the most commonly encountered complications. The pathogenesis for the development of colitis is unclear, and there is a growing consensus that the ecosystem of the gastrointestinal microbiota plays a significant role. Based on this suspected connection, studies are being carried out to explore the changes in the microbiota in patients on these medications who develop colitis. Conceivably, the modulation of the gut microbiota could offer a therapeutic benefit. Fecal microbiota transplantation is one therapeutic option that is currently being investigated, though there are still more data needed to evaluate its efficacy. In this review, we recapitulate the mechanisms of action of immune checkpoint inhibitors, their adverse events, with a focus on colitis and the role gut microbiota are suspected to play, and finally discuss the microbiota modulation therapies being investigated.
Immune checkpoint inhibitors (ICIs) are a revolutionary class of drugs used as cancer immunotherapy, particularly known to prolong the survival in patients with metastatic melanoma, lung cancer, bladder cancer and many others.1 These agents act by targeting programmed death protein-1 (PD-1) or its associated ligand (PDL-1) or cytotoxic T-lymphocyte-associated protein-4 (CTLA-4),2 all of which are immunological down-regulators. The PD-1 receptor is expressed on the surface of monocytes, T cells, B cells, dendritic cells and tumor-infiltrating lymphocytes.1 Its ligand, PDL-1 is often present in abundance on tumor cells and antigen presenting cells, which after binding with PD-1, leads to suppression of T-cell receptor signaling.1 Similarly, CTLA-4 is a receptor present on the surface of CD4+ and CD8+ T cells, subsets of B cells and thymocytes.3 It is responsible for the inhibition of T-cell activation at the initial step in an immune response.4 Antibodies against these immune checkpoints have been utilized to strengthen the immune response to increase the overall survival for cancer patients. Ipilimumab was the first immunotherapeutic agent to be approved by the Food and Drug Administration in 2011 primarily for the management of non-resectable melanoma.5 Ipilimumab is a human monoclonal antibody (immunoglobulin G1 [IgG1]) that targets cytotoxic T-lymphocyte antigen. Since then, a plethora of drugs for targeted immunotherapy have developed that function via both pathways, including nivolumab, pembrolizumab, atezolizumab and many others, that are PD-1/PDL-1 inhibitors.6 However, due to their action of mass activation of T-cells that can infiltrate nearly any organ in the body, it is essential to monitor for the toxicities associated with their use. Increasing immune system activity by immune checkpoint blockade can result in inflammatory side-effects, known as immune-related adverse events (irAEs).4
irAEs associated with ICIs are commonly known to affect the colon, liver, lungs, pituitary, thyroid and skin, and less commonly the heart, nervous system and other organs.7 The reported incidence of irAEs from different clinical trials has been variable, largely due to an inconsistent definition of an irAE. An irAE incidence of 15% to 90% (of any grade) has been reported and severe toxicities requiring immunosuppression after discontinuation of the offending drug occurred 0.5% to 13% of the time.8 Most of these clinical trials state that gastrointestinal tract toxicities tend to be the most commonly occurring serious irAEs.9 A systematic review conducted by Wang et al.7 focused on fatal toxicities of all ICI therapies using the database of the World Health Organization, reported a total of 613 fatal events from 2009 to 2018. Out of these, 198 deaths were due to anti-CTLA-4 therapy, and colitis was the cause in 135 cases (70%).7
Numerous mechanisms have been proposed for irAEs that occur with ICI usage. One belief is that since CTLA-4 is responsible for the regulation of self-antigen tolerance, blockage of CTLA-4 can lead to downstream autoimmunity leading to CD4 T-helper cell and CD8 T-cell tissue infiltration and cytokine release.10 Mouse modes of regulatory T-cells (Treg) deficiency subsequently developed autoimmune diseases.11 Since CTLA-4 is abundantly expressed on the surface of Treg cells, CTLA-4 blockade could lead to diminished numbers of Treg and lead to autoimmunity. Recent data advocate the involvement of human gastrointestinal microbiota in the development of ICI induced colitis.
There appears to be an emerging role of the human gastrointestinal microbiota in the pathogenesis of ICI induced colitis. A prospective study of patients with metastatic melanoma treated with ipilimumab was carried out to establish an association between baseline gut microbiota and the response to ipilimumab and subsequent development of colitis.12 Fecal microbiome analysis was performed at baseline and before each infusion. Patients who had gut microbiota enriched with Faecalibacterium and other Firmicutes had a better overall survival despite an association with the development of colitis as an irAE, as compared to patients with microbiota enriched with Bacteroidetes.12
Another study by Dubin et al.13 sought to characterize the gut microbiota before the development of colitis in patients and identify biomarkers that may indicate relative protection against CTLA-4 inhibition induced colitis. Colitis-resistant patients had greater numbers of Bacteroidetes, which may have limited inflammation by supporting differentiating pathway to a Treg phenotype. They further studied metabolomics and suggested that the presence of bacterial polyamine transport system and thiamine, riboflavin and pantothenate be used to assess the risk of developing colitis as these vitamins are found to be low in patients with inflammatory bowel disease.13 Polyamines (amines that are exported from cells of bacteria via specific transport systems) are believed to maintain the colonic epithelial barrier by encouraging proliferation and the enzymes involved in the synthesis of these polyamines are lower in patients with colitis.13 Subsequent data gathered from various other studies similarly point towards the suspected association of gut microbiota with the response to ICI therapy as well as the potential development of enterocolitis.14,15
Interestingly, another study demonstrated that the use of antibiotics had a negative impact of the survival of patients on PDL-1 therapy.16 Antibiotics given after the initiation of ICI therapy resulted in a more severe disease course and a shorter overall survival. These findings further consolidate the evidence of a connection between the gut microbiota and its impact on the effectiveness of ICI therapy and potential irAEs.
Immunotherapy-induced colitis often presents with nonspecific symptoms such as abdominal pain and diarrhea or sometimes with severe symptoms such as blood in stool or ileus.17 Guidelines for management of new-onset diarrhea in a patient on an ICI regimen recommend ruling out other infectious causes including Clostridioides difficile, along with other enteric pathogens.18 Imaging in the form of an abdominal computed tomography may be used to diagnose colitis.6 For more severe colitis, endoscopy with biopsies should be taken for a definite diagnosis.6
In a study by Marthey et al.19 endoscopic findings of the colon in patients undergoing anti-CTLA-4 therapy included ulcers, erosions and erythema. The rectum and/or sigmoid colon were involved in 97% of the cases and that lesions were patchy in 55% of the patients. Other lesions usually seen on endoscopy include exudates, granularity and loss of vascular pattern, which appear to be similar to those seen in inflammatory bowel disease (ulcerative colitis and CrohnŌĆÖs disease).20
In addition to endoscopic findings, histological findings in ICI induced colitis are also similar to inflammatory bowel disease. On histology, intraepithelial neutrophils in the surface epithelium and crypt epithelium are seen in ICI induced colitis similar to inflammatory bowel disease.21 Crypt irregularities and crypt abscesses, which are features considered to be classically present in inflammatory bowel disease, are also seen in ICI induced colitis.
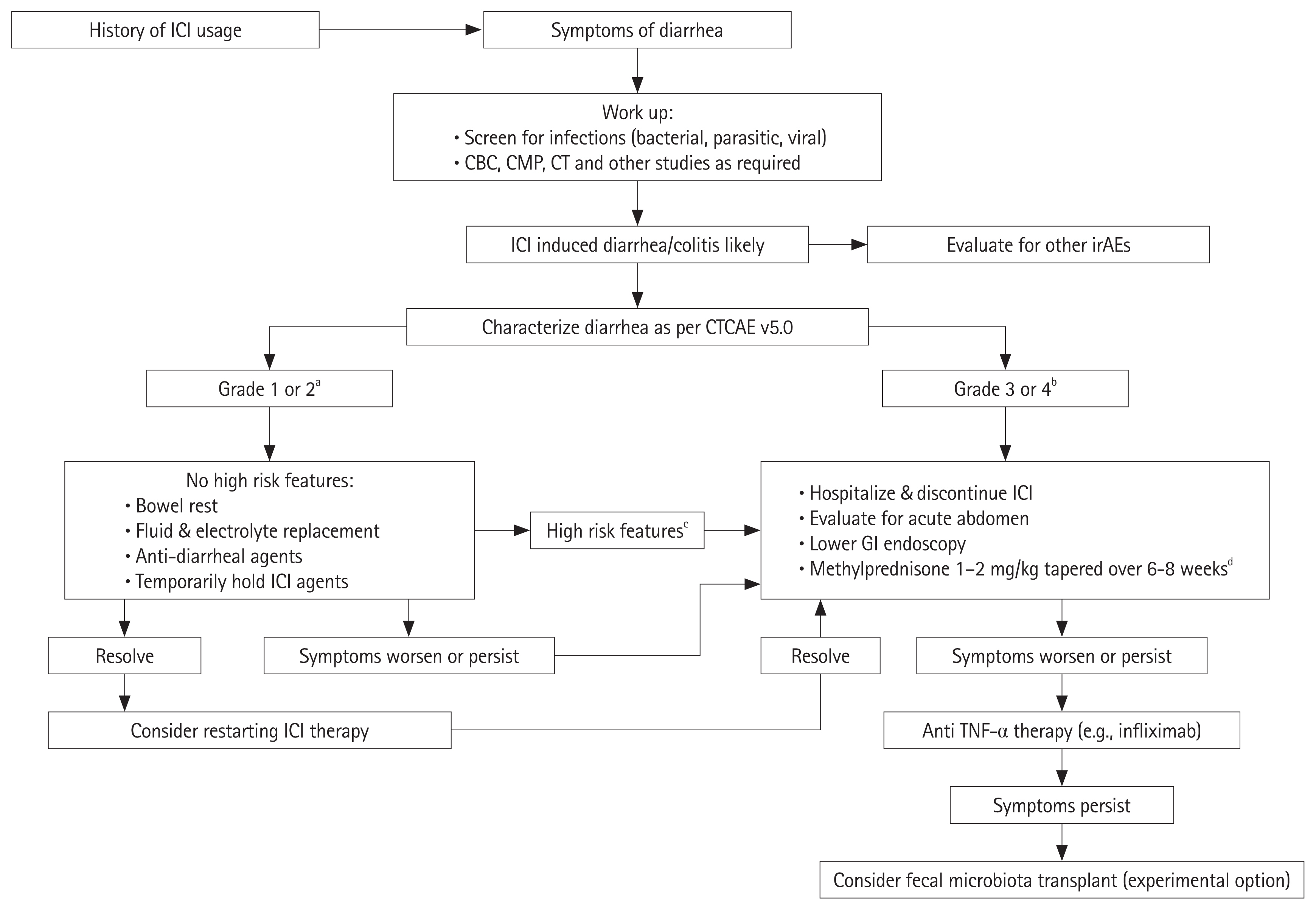
When colitis in a patient is suspected to be an irAE, treatment for the condition usually varies according to the severity/grade of the colitis,3 using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 5.0.22 We have summarized an approach to the management of ICI induced colitis in Fig. 1. Lower grade diarrhea can be managed by using a low dose corticosteroid taper and continuing ICI therapy can be considered, based on individual patient requirements, while higher grade diarrhea or diarrhea symptoms that persist or worsen despite corticosteroid treatment need to permanently discontinue ICI therapy and higher doses of systemic corticosteroids.3 If despite a higher dose of corticosteroids, symptoms worsen or persist, anti-tumor necrosis factor-╬▒ therapy (such as infliximab) can be administered.3
With the suspected interplay between the gut microbiota and the potential development of colitis by ICIs, recent exploration of therapeutic advances have led to the conjecture that microbiota modulation therapies could be utilized for colitis. One such option being investigated is fecal microbiota transplant (FMT).
A case series by Wang et al.23 included 2 patients that were suffering from refractory CTLA-4 induced colitis. The first patient was diagnosed with metastatic urothelial carcinoma nonresponsive to standard chemotherapy and received a trial of CTLA-4 and PD-1 blockade, subsequently developed CTCAE grade Ōēź2 diarrhea/colitis. As treatment for the resultant colitis, the patient received systemic corticosteroids, 2 doses of infliximab and vedolizumab but had no settlement of her symptoms. The second patient had prostate cancer nonresponsive to chemotherapy as well as hormonal therapy, and was started on ipilimumab as a part of a clinical trial. However shortly after, the patient developed fever and CTCAE grade Ōēź2 diarrhea/colitis. Despite receiving therapy with systemic corticosteroids, infliximab and vedolizumab, symptoms persisted. Both these patients subsequently received FMT from unrelated healthy donors and had complete resolution of their symptoms. They also analyzed the gut microbiota composition of the 2 patients before and after the FMT and demonstrated that the bacterial taxa of the 2 patients and the donor were different at baseline. Prior to FMT there was mainly an abundance of Clostridia and lack of Bacteroidetes and Verrucomicrobia (which are considered to be protective from ICI colitis and inflammatory bowel disease respectively)12,13 in the first patient, while the second patient had Escherichia (linked with gut microbiome dysbiosis).23 They further reported that immediately after FMT the first patient had an increase in Akkermansia (but this change was short lived), with an expansion of Bifidobacterium which occurred after 7 weeks of transplant, while this expansion was seen immediately in the second patient. Studies conducted in mice have found that Bifidobacterium protects against irAEs from ICI and reduces intestinal inflammation.24 Similarly, another case report of a patient suffering from metastatic gastric adenocarcinoma developed symptoms suggestive of ICI colitis after initiating treatment with pembrolizumab. The resultant colitis was nonresponsive to treatment with a plethora of drugs including intravenous steroids, infliximab, mycophenolate mofetil, mesalamine, vedolizumab, diphenoxylate, atropine, and loperamide. Eventually a decision was made to initiate FMT as a mode of treatment for the patient, after which the patient had a remarkable improvement of his symptoms.25
This recent realization further raises the question of the utility of microbial therapies to manipulate the gut microbiota ecosystem in order to prevent or even treat the colitis that develops due to the use of ICIs. According to clinicaltrials.gov, there are 2 clinical trials being planned use FMT for management of ICI adverse events (NCT04038619 and NCT03819296).
ICIs have changed the face of treatment for a number of cancers by successfully prolonging the survival of patients. But, their increasing use has proven to be a double-edged sword. Owing to their immunologic mechanism of action, the consequent adverse events can affect any organ in the body. Colitis has been increasingly seen as one of the most common and most problematic event. Even though the exact pathogenesis of the condition is unclear, there is a growing body of evidence that gastrointestinal microbiota has an impact on the subsequent development of colitis with ICI use. A number of studies have explored the manipulation of the gut ecosystem for the potential treatment of colitis in the form of FMT which has thus far exhibited promising results. However, more data need to be gathered to establish the beneficial effect of FMT as a treatment option and also as a therapy for relapse. Further, more studies are required to verify the exact bacterial strains that influence colitis development, including distinguishing strains that have a protective effect.
ADDITIONAL INFORMATION
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Fig.┬Ā1
Management algorithm for ICI-induced colitis. aGrade 1: increase of <4 stools/day from baseline and grade 2: increase of 4-6 stools/day from baseline; bGrade 3: increase of Ōēź7 stools/day; hospitalization indicated and grade 4: life threatening consequences; urgent intervention indicated; cHigh risk features: grade 1 or 2 diarrhea or grade 3 or 4 diarrhea with the following signs or symptoms: moderate to severe cramping, decreased performance status, fever, sepsis, neutropenia, frank bleeding or dehydration23; dIntravenous methyl-prednisone is first line therapy for severe diarrhea until stabilization (3 days to 2 weeks) followed by oral prednisone dose tapered over 6-8 weeks.3 ICI, immune checkpoint inhibitor; CBC, complete blood count; CMP, comprehensive metabolic panel; irAEs, immune-related adverse events; CT, computed tomography; CTCAE v5.0, Common Terminology Criteria for Adverse Events: version 5.0 for diarrhea and colitis22; GI, gastrointestinal; TNF, tumor necrosis factor.
REFERENCES
1. Reddy HG, Schneider BJ, Tai AW. Immune checkpoint inhibitor-associated colitis and hepatitis. Clin Transl Gastroenterol 2018;9:180.



2. Abu-Sbeih H, Wang Y. Gut microbiome and immune checkpoint inhibitor-induced enterocolitis. Dig Dis Sci 2020;65:797-799.


3. Gupta A, De Felice KM, Loftus EV Jr, Khanna S. Systematic review: colitis associated with anti-CTLA-4 therapy. Aliment Pharmacol Ther 2015;42:406-417.


4. Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med 2018;378:158-168.


5. Lacroix M. Targeted therapies in cancer. Berlin: Springer, 2014.
6. Singh BP, Marshall JL, He AR. Workup and management of immune-mediated colitis in patients treated with immune checkpoint inhibitors. Oncologist 2020;25:197-202.


7. Wang DY, Salem JE, Cohen JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol 2018;4:1721-1728.



8. Kumar V, Chaudhary N, Garg M, Floudas CS, Soni P, Chandra AB. current diagnosis and management of immune related adverse events (irAEs) induced by immune checkpoint inhibitor therapy. Front Pharmacol 2017;8:49.



9. Michot JM, Bigenwald C, Champiat S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer 2016;54:139-148.


10. Som A, Mandaliya R, Alsaadi D, et al. Immune checkpoint inhibitor-induced colitis: a comprehensive review. World J Clin Cases 2019;7:405-418.



11. Read S, Malmstr├Čm V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25(+)CD4(+) regulatory cells that control intestinal inflammation. J Exp Med 2000;192:295-302.



12. Chaput N, Lepage P, Coutzac C, et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann Oncol 2019;30:2012.


13. Dubin K, Callahan MK, Ren B, et al. Intestinal microbiome analyses identify melanoma patients at risk for checkpoint-blockade-induced colitis. Nat Commun 2016;7:10391.



14. Dutta D, Lim SH. Bidirectional interaction between intestinal microbiome and cancer: opportunities for therapeutic interventions. Biomark Res 2020;8:31.



15. Salgia NJ, Bergerot PG, Maia MC, et al. Stool microbiome profiling of patients with metastatic renal cell carcinoma receiving anti-PD-1 immune checkpoint inhibitors. Eur Urol 2020;78:498-502.


16. Routy B, Gopalakrishnan V, Daill├©re R, Zitvogel L, Wargo JA, Kroemer G. The gut microbiota influences anticancer immunosurveillance and general health. Nat Rev Clin Oncol 2018;15:382-396.


17. Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the immune-related adverse effects of immune checkpoint inhibitors: a review. JAMA Oncol 2016;2:1346-1353.


18. Abu-Sbeih H, Ali FS, Wang Y. Immune-checkpoint inhibitors induced diarrhea and colitis: a review of incidence, pathogenesis and management. Curr Opin Gastroenterol 2020;36:25-32.


19. Marthey L, Mateus C, Mussini C, et al. Cancer immunotherapy with anti-CTLA-4 monoclonal antibodies induces an inflammatory bowel disease. J Crohns Colitis 2016;10:395-401.



20. Iranzo I, Huguet JM, Su├Īrez P, Ferrer-Barcel├│ L, Iranzo V, Sempere J. Endoscopic evaluation of immunotherapy-induced gastrointestinal toxicity. World J Gastrointest Endosc 2018;10:392-399.



21. Verschuren EC, van den Eertwegh AJ, Wonders J, et al. Clinical, endoscopic, and histologic characteristics of ipilimumab-associated colitis. Clin Gastroenterol Hepatol 2016;14:836-842.


22. Common terminology criteria for adverse events (CTCAE) version 5.0. National Cancer Institute Web site
https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htmUpdated November 27, 2017. Accessed November 10, 2020
23. Wang Y, Wiesnoski DH, Helmink BA, et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor-associated colitis. Nat Med 2018;24:1804-1808.











