Very early onset inflammatory bowel disease in a South Asian country where inflammatory bowel disease is emerging: a distinct clinical phenotype from later onset disease
Article information
Abstract
Background/Aims
Information on pediatric inflammatory bowel disease (PIBD) and very early onset IBD (VEOIBD) are sparse in India, where IBD is emerging. We aimed to evaluate characteristics of VEOIBD and later onset PIBD (LO-PIBD) in India.
Methods
We performed retrospective analysis of a large, prospectively maintained IBD registry. PIBD was divided in to VEOIBD (< 6 years) and LO-PIBD (6–17 years). Demographic data, disease characteristics and treatment were compared between the PIBD groups and with other Asian/Western studies as well as the adult patients of the registry.
Results
Of 3,752 IBD patients, 292 (7.8%) had PIBD (0–17 years) (175 Crohn’s disease [CD], 113 ulcerative colitis [UC], 4 IBD-undifferentiated; 22 VEOIBD [7.5%], and 270 LO-PIBD [92.5%]). VEOIBD patients had more severe disease compared to LO-PIBD in both UC (P=0.003) and CD (P<0.001). Familial IBD was more common in VEOIBD (13.6%) compared to LO-PIBD (9.2%). Ileal disease (L1) was an independent risk factor for diagnostic delay in pediatric CD. Diagnostic delay (> 6 months) was significantly lower in VEOIBD (40.9%) than in LO-PIBD (78.8%) (P<0.001). Compared to other Asian and Western studies, extensive UC (72.5%) and complicated CD (stricturing/penetrating: 42.7%) were relatively more common. Perianal CD was relatively less frequent (7.4%). PIBD had a significantly higher number of complicated and ileal CD and extensive UC comparison to adult cohort of the registry.
Conclusions
VEOIBD has more aggressive phenotype than LO-PIBD. Disease appears distinct from other Asian and Western studies and adult onset disease, with more complicated CD and extensive UC.
INTRODUCTION
Pediatric inflammatory bowel disease (PIBD; age < 17 years) constitutes up to 10%–25% of all IBD patients [1]. Of these, a small proportion are very early onset IBD (VEOIBD; < 6 years). VEOIBD appears to be a distinct subset of pediatric IBD with unique epidemiological and phenotypic characteristics including a severe and aggressive course of disease, predominant colonic involvement and a higher incidence of familial IBD compared to PIBD other than VEOIBD (6–17 years) [1].
The incidence of PIBD is on the rise globally [2]. A complex interplay of genetic and changing environmental factors has been implicated for the rising incidence although specific factors are yet to be completely understood [2-6].
Most of the epidemiological data however are from the West including North America, Canada, and Europe [3]. A few recent studies from Asia also suggest a rising trend of PIBD. Interestingly the phenotype appears to be distinct from that of the West including a milder ulcerative colitis (UC) and a higher perianal involvement in Crohn’s disease (CD) [4-10]. India is a large country with a large population where IBD is a new but emerging disease. Though the incidence of IBD is lower than that of the West expectedly, the burden of disease is high. The diagnosis of IBD itself poses significant challenges in a country where infectious diseases predominate, and tuberculosis (TB) is endemic [11,12].
Data on PIBD behavior and outcomes from India is limited. There are no published reports or separate analysis of VEOIBD and its differences from later onset disease. Similarities to adult IBD and distinctive features have been described in small and questionnaire-based studies [5,13-20].
The primary aim of this study was to evaluate the disease characteristics and treatment patterns in VEOIBD and compare that to later onset PIBD (LO-PIBD; 6–17 years). We also compared our data with other PIBD or VEOIBD studies from Asia and the West.
METHODS
1. Study Population
The study was conducted at the Asian Institute of Gastroenterology (AIG) in Hyderabad, India, a gastroenterology tertiary center for IBD patients across India. The IBD center was established in 2004 with a well-established prospectively maintained registry [21].
The registry includes the detailed history and demographic profile since the first visit, symptoms, disease activity scores, medication, and family history collected by interview and chart review. Clinical characteristics, including family history and response to treatment, are updated at each follow-up visit [12,22].
Eligible subjects included all patients with confirmed IBD who were less than 17 years of age up to November 2019. All patients underwent at least one ileo-colonoscopy and gastroscopy before enrolment into the registry. Patients with a follow-up of less than 6 months were excluded from the study.
The diagnosis of IBD was based on the European Society for Pediatric Gastroenterology Hepatology and Nutrition (ESPGHAN) “Porto” criteria including history, clinical, endoscopic, histologic and radiologic features [23]. Small bowel evaluation was done by magnetic resonance enterography in majority of the cases. Computed tomography enterography was used in very limited numbers due to risk of radiation. A chest X-ray, 2 step Mantoux and biopsy specimen polymerase chain reaction for Mycobacterium. TB DNA were done to rule out TB in CD patients. Clinical, endoscopic, radiologic features and histopathology were considered for differentiating intestinal TB from CD. Empirical anti-TB therapy (ATT) was initiated when diagnostic dilemma persisted even after extensive evaluation. An assessment for response is done at 2 months. In case of improvement, ATT is continued for at least 6 months.
The patients were classified as VEOIBD < 6 years of age, and LO-PIBD for patients between 6 and 17 years of age. The subgroup of patients less than 2 years of age were identified as infantile onset IBD (IOIBD). Demographic data including age, sex, family history, clinical symptoms at presentation (abdominal pain, diarrhea, weight loss, and rectal bleeding) and presence of extraintestinal manifestations was retrieved from the registry. The diagnostic delay was calculated based on the difference between the age of onset of symptoms and the age at diagnosis.
2. Disease Extension and Severity
The location of the disease was noted using the pediatric modification of Montreal classification (Paris classification) for the purpose of the study [24]. The disease activity at presentation was recorded according to the Pediatric Crohn’s Disease Activity Index (PCDAI) and the Pediatric Ulcerative Colitis Activity Index (PUCAI) [25,26]. The maximum extent recorded and the maximum severity noted were included in the analysis.
The treatment given including the requirement of steroids, immunomodulators and biologics was presence of complications and need for surgeries were recorded.
3. Statistical Methods
The IBD registry runs on Apex healthcare management system software which allows entry of follow-up data at each visit. The data for the PIBD and VEOIBD patients were reviewed for completeness and consistency before entering into Microsoft Excel (Microsoft Corp., Redmond, WA, USA).
Continuous variables were expressed as mean (95% confidence interval [CI]) if normally distributed or as median and interquartile range (IQR)/range if not normally distributed. Normality of distribution was ascertained by the Kolmogorov-Smirnov test and the Shapiro-Wilk test (P<0.05 indicates nonnormal distribution). Categorical variables were expressed as frequencies and percentages. Patient characteristics were compared using Student t-test (for data with normal distribution) and Mann-Whitney test (for non-parametric data/data with non-normal distribution with 2 groups) for continuous variables and chi-square/Fisher exact test (depending on sample size and expected values) for categorical variables. Univariate followed by multivariate logistic regression analysis was performed to look for risk factors of diagnostic delay ( > 50th percentile) in CD and UC and also to assess risk factors for surgery in CD. P-values of < 0.05 were considered statistically significant. The data were analyzed using SPSS version 21.0 (IBM Corp., Armonk, NY, USA).
4. Ethical Considerations
All patients seen at the clinic with a confirmed diagnosis of IBD are enrolled into the registry after informed consent including permission to use information for research purposes. For pediatric cases (age of onset < 17 years) informed consent is obtained from the parent or legal guardian. The study was approved by the Institutional Review Board of Asian Institute of Gastroenterology (AIG AHF IRB No. 07/2019) and performed in accordance with the principles of the Declaration of Helsinki.
RESULTS
1. Demographics and Disease Presentation
The AIG IBD registry included 3,752 patients. Of these, 292 subjects (7.8%) comprised the PIBD cohort (age < 17 years). Majority (n = 270, 92.5%) of the PIBD cases belonged to the LO-PIBD group (6–17 years). VEOIBD ( < 6 years) and IOIBD ( < 2 years) were identified in 22 (7.5%) and 8 (2.7%) children, respectively. The distribution of the pediatric cases according to age is shown in Fig. 1A.

Age distribution, disease extent, and phenotype of PIBD. (A) Distribution of UC and CD cases according to age groups. (B) Extent of disease in UC: VEOIBD and LO-PIBD patients. (C) Extent of disease in CD: VEOIBD and LO-PIBD patients. (D) Disease behavior in CD: VEOIBD and LO-PIBD patients. IBD, inflammatory bowel disease; CD, Crohn’s disease; UC, ulcerative colitis; IBD-U, undifferentiated IBD; VEOIBD, very early onset IBD; LO-PIBD, later onset pediatric IBD; E1, proctitis; E2, upto splenic flexure; E3, upto hepatic flexure; E4, pancolitis; L1, ileal; L2, colonic; L3, ileo-colonic; L4a, upper gastrointestinal disease upto ligament of Treitz; L4b, beyond ligament of Treitz upto distal ileum CD; B1, inflammatory; B2, stricturing disease; B3, fistulizing CD; p, perianal disease.
There were more cases of CD (59.9%, 175/292) than UC (38.7%, 113/292) in all the age groups. IBD-undifferentiated (IBD-U) was seen in 4 patients (1.4%). CD was diagnosed in 13 (59.1%) and UC in 8 (36.3%) of VEOIBD cases. IBD-U was seen in 1 patient (4.5%) of VEOIBD group.
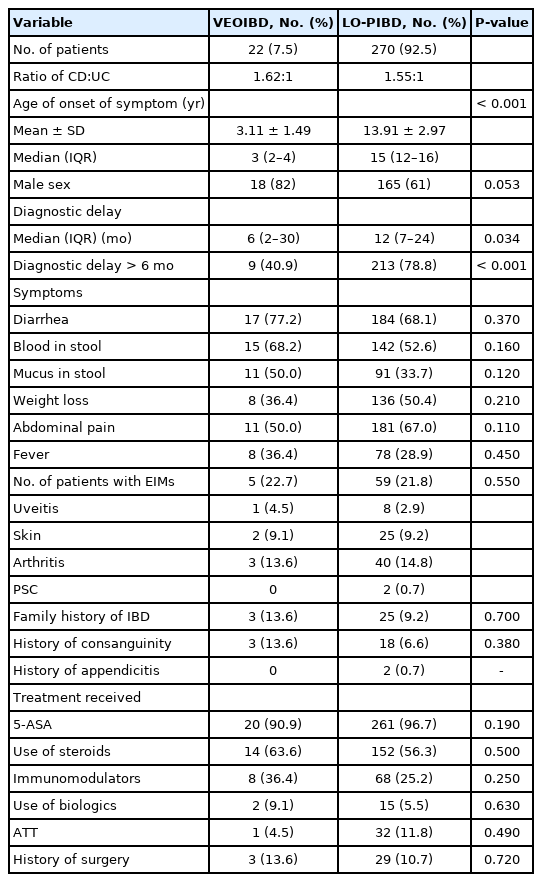
The presenting symptoms of VEOIBD are tabulated in Tables 1 and 2. There were no significant differences in the symptoms at onset, presence of extraintestinal manifestations and treatment received between VEOIBD and LO-PIBD (Tables 1, 2).
2. Family History
A family history of IBD was noted in 3 (13.6%) of VEOIBD and 25 (9.2%) children with LO-PIBD (P=0.70). Three (13.6%) of VEOIBD and 18 (6.6%) of LO-PIBD were born of consanguineous marriage (P=0.38).
3. Delay in Diagnosis
Diagnostic delay (> 6 months) was significantly lower in VEOIBD (40.9%) than in LO-PIBD (78.8%) (P<0.001). The median diagnostic delay in VEOIBD and LO-PIBD were 6 months (IQR, 2–30 months) and 12 months (IQR, 7–24 months), respectively (P=0.034). On further analysis, there was no significant difference in diagnostic delays between UC (median, 18 months; IQR, 3–29 months) and CD (median, 20 months; IQR, 6–30 months) in each group (P=0.30).
4. Risk Factors for Diagnostic Delay
Logistic regression analyses of risk factors for diagnostic delays ( > 50th percentile) in the diagnosis of CD and UC are depicted in Table 3. In CD, ileal disease was a risk factor for diagnostic delay (> 50th percentile) (P=0.03; odds ratio [OR], 2.16). In UC, none of the factors were predictive except age > 10 years (which nearly achieved significance: P=0.06; OR, 0.21).
5. ATT Prior to Diagnosis
A total of 31 children (17.7%; 2 in VEOIBD, 29 in LO-PIBD) of pediatric CD received ATT prior to diagnosis. Pediatric UC patients of 1.8% (2/113) received ATT. Empirical ATT use was lower in VEOIBD compared to LO-PIBD, but not statistically significant (P=0.49).
6. Disease Extent and Severity
Fig. 1B and C show the extent of disease in UC and CD in VEOIBD and LO-PIBD patients.
1) Ulcerative Colitis
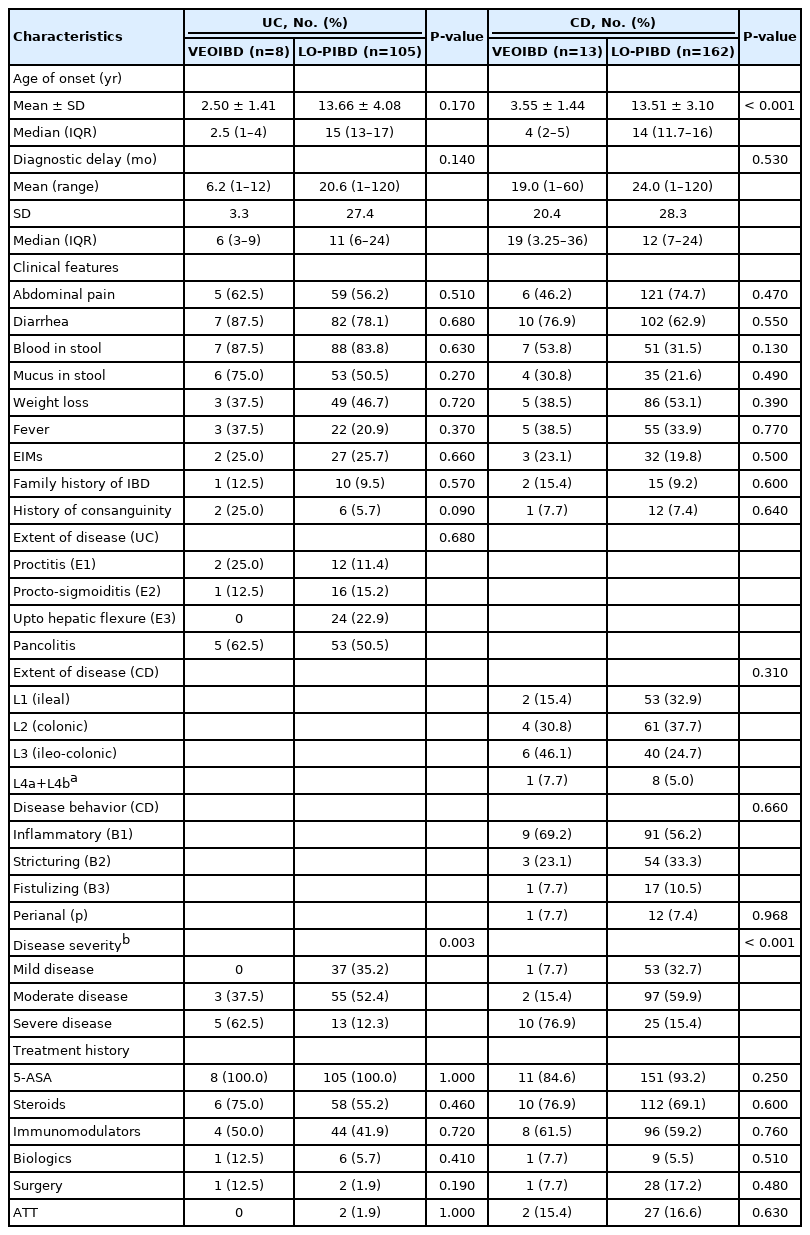
A higher proportion of patients with VEOIBD had extensive colitis compared to LO-PIBD (62.5% vs. 50.5%). However, the difference was not statistically significant (P=0.68) (Fig. 1B). Similarly, the severity of disease was significantly higher for VEOIBD than LO-PIBD (Table 2). The median PUCAI score for VEOIBD was 60 (range, 45–80). The severity of disease according to the PUCAI score was moderate and severe in 37.5% and 62.5%, respectively with no mild cases. This was significantly higher than in LO-PIBD where 35% had mild disease (Table 2).
2) Crohn’s Disease
Ileo-colonic involvement (L3) was more common in VEOIBD group than LO-PIBD patients. Ileal and colonic diseases were more common in LO-PIBD (Fig. 1C). However, none of the differences were statistically significant (0.31). Inflammatory disease phenotype (B1) was more common in both VEOIBD and LO-PIBD group in CD. Stricturing (B2) and fistulizing (B3) diseases were more common in LO-PIBD, but no statistically significant difference was noted (P=0.66). Perianal was found in similar proportion of patients in both subgroups (7.7% in VEOIBD group and 7.4% in LO-PIBD group) (P=0.97) (Fig. 1D). The median PCDAI score for VEOIBD was 47.5. Mild, moderate, and severe disease were found in 7.5%, 15.4%, and 76.9 % of children, respectively. This was significantly higher than the LO-PIBD group (Table 2).
7. Infantile Onset IBD (< 2 Years)
IOIBD ( < 2 years at onset of symptoms) was noted in 8 children (2.7%) of PIBD. Of these, 3 (37.5%) had complications with an aggressive disease course. The first child with CD was symptomatic for 1 month of age, with colonic ulcerations and extensive skin excoriations. His elder sibling had died of similar complaints at 1.5 years of age. Genetic testing in this child revealed interleukin (IL)-10 receptor mutations. He underwent a bone marrow transplant and was better at 8 months post-transplant. The second child required hemicolectomy for severe unresponsive disease at 6 years of age. The third child with UC was lost to follow-up for more than 2 years and expired at 5 years of age presumably due to septicemia. The 5 other cases with IOIBD also had a severe disease course with multiple relapses needing steroids.
8. Treatment Received
There was no significant difference in the use of biologics, steroids, 5-aminosalicylic acid (5-ASA), and immune modulators (azathioprine/methotrexate) between VEOIBD and LO-PIBD (in total as well as in subgroups of UC and CD there was no significant difference) (Table 2).
All patients with UC received 5-ASA. Sixty-four out of 113 patients (56.6%) required a course of steroids. Steroid dependent and refractory UC were put on immunomodulators to maintain remission (42.4%, 48/113). Eleven out of 113 patients with UC (9.7%) had frequent relapsing course of disease ( > 3 relapses a year) and 3 patients had acute severe colitis requiring surgery (2.65%). Fifteen out of 175 CD patients (8.5%) had > 3 relapses in a year. Steroid and azathioprine requirements were higher in CD but biologic usage was still low (5.7%, 10/175) (Table 2). Thirty-seven out of 175 CD patients (21.1%) received partial enteral nutrition (PEN) (approximately half of the daily calorie requirement) in addition to other therapy.
Surgery was required in 3 of 22 (13.6%) of VEOIBD and 29 of 270 (10.7%) LO-PIBD children. These included proctocolectomy (1), sub-total colectomy (3), right hemicolectomy (1), segmental small bowel resection (12 ileal, 6 jejunal) and ileocecal resection (5). Fistulectomy and examination under anesthesia with seton placement was done in 4 patients. 16.5% (29/175) patients with CD and 2.4% (3/114) patients with UC underwent surgery. Logistic regression analysis was done to assess for risk factors of surgery–none of the risk factors significantly predicted surgery (Supplementary Table 1). There were 4 teenage smokers in our pediatric cohort (3 CD and 1 UC).
DISCUSSION
This is a retrospective analysis of prospectively maintained database of a large number of PIBD patients. This is the first study from India that has highlighted the disease characteristics and treatment patterns of PIBD particularly VEOIBD and IOIBD and compared the differences with later onset disease (6–17 years). Severity of disease was significantly higher in VEOIBD (in both UC and CD subgroups) compared to later onset disease (P=0.003 and P<0.001, respectively) consistent with earlier studies [27,28].
A family history of IBD was found in 14% patients with VEOIBD compared to 9% in later onset disease. The entire cohort from our registry has a positive family history in only 4% (CD of 4.13% and UC of 4.34%) which we have published recently [29]. This has been reported in many other studies that familial IBD is less common in Asia than the West [30,31]. This clearly demonstrates that a genetic predisposition to IBD correlates with the onset of disease being highest in the IOIBD and VEOIBD.
We compared our data with that of other studies both in Asia and the West (Supplementary Tables 2, 3). The large Western population-based Epidemiology of inflammatory bowel diseases: new insights from a French population-based registry (EPIMAD) (1,412 PIBD patients) was used for comparison (Supplementary Table 2) [32]. Male preponderance was more evident (82% vs. 52%, P=0.02). The VEOIBD disease character otherwise was similar to our study. Interestingly the LO-PIBD group in our study had significant differences compared to EPIMAD study. A higher number of patients had a diagnostic delay > 6 months (78.8% vs. 30%, P=0.004) and the incidence of familial IBD (9.2% vs. 15%, P=0.009) was significantly lower. The Indian PIBD appears to be more severe with a higher incidence of stricturing and penetrating disease (B2 and B3) in CD and pan colitis (E4) in UC compared to EPIMAD cohort [31].
We also compared our data with other PIBD cohorts from Asia and Europe (Supplementary Table 3) [6,7,16,17,32]. We noted a high incidence of ileal CD (31.4%) though colonic disease was most common (37.1%) [10]. Another Indian study by Sonavane et al. [17] similarly showed that the most common phenotype was ileal (37.5%). Perianal disease and upper gastrointestinal (L4) was less common. A recent study on PIBD from East Asia (Japan) showed higher incidence of upper gastrointestinal and perianal disease compared to European Cohort (EUROKIDS) [32,33]. The incidence of structuring and fistulizing disease was significantly higher than other studies. The rates of surgery in CD (16.5%) comparable to other Asian study (South Korea, 22.7%) [34,35]. In UC, although pan colitis was the most common phenotype, colectomy rates were lower (2.7%) supporting the notion that UC is a milder disease in Asian countries with less incidence of acute severe colitis and colectomy (Supplementary Table 3) [13,16,36,37]. Compared to another questionnaire-based survey from India by Sathiyasekaran et al. [16], the proportion of VEOIBD, disease location and proportion of perianal CD were different. Such differences could be due to variation in methods of data collection and study settings [22]. However, results appear more consistent with a recent study from India by Sonavane et al. [17].
We found that a diagnostic delay > 6 months was lesser in VEOIBD compared to later onset disease (40.9% vs. 78.8%, P<0.001) disease perhaps because of more severe disease at presentation prompting early referral in VEOIBD. Another plausible explanation could be the use of diapers in infants and hence easier observation of their stools. Ileal disease (L1) was predictive of longer diagnostic delay ( > 50th percentile) in CD [38]. This is in sharp contrast to the EPIMAD study where diagnostic delay > 6 months were seen in only 27% and 30% patients respectively in VEOIBD and LO-PIBD [32]. The disproportionately longer delay in diagnosis could be attributable to poor physician awareness of pediatric IBD in India and high percentage of ileal (31.4%) disease in our study which is a known risk factor for diagnostic delay in pediatric CD [39].
The other contributory factors for diagnostic delay include the infectious mimics of IBD and over the counter antibiotics (like fluoroquinolone and metronidazole) are easily available and is often the first line of treatment. The high prevalence of TB which is often symptomatically, endoscopically and histopathologically difficult to differentiate from ileo-colonic CD is another diagnostic dilemma which results in empirical use of ATT [39-42]. Almost one-fifth of children with CD in our cohort had received ATT prior to diagnosis adding to diagnostic delay. We have earlier reported that empirical use of ATT was the single largest contributor for diagnostic delay in an adult CD cohort of more than 700 patients which resulted in higher long term stenotic complications and need for surgery [43]. India is endemic for TB and there is limited awareness about CD. Hence at the community level, patients are initiated on ATT and CD is diagnosed only if there is no response leading to inordinate delays in diagnosis.
We also compared data between the adult and PIBD cohort in our registry (Supplementary Table 4). Familial IBD was more common in PIBD cohort. Higher incidence of ileal disease (L1), stricturing phenotype (B2) and perianal disease was found in pediatric CD cohort. UC in PIBD cohort was more extensive. Steroid, immunomodulator, and biologic usage was also higher in PIBD group. However, the diagnostic delay was significantly higher (P<0.001) in adults with CD (mean ± standard deviation [SD], 42.2 ± 46.1 months) compared to pediatric CD (mean ± SD, 23.56 ± 27.76 months) [22]. This could be attributable to the higher usage of empirical ATT in the adult cohort.
IOIBD was seen in 8 children (2.7% of PIBD). Three had refractory disease requiring surgery. IL-10 receptor mutation was detected in 1 child who later underwent allogenic hematopoietic stem cell transplantation. This is very much in concordance with other studies from across the globe suggesting that IOIBD is a distinct and the most difficult to treat subset of pediatric IBD [44]. Monogenic defects like IL-10/IL-10R mutations are not uncommon in this subgroup [45,46]. However, mutational defects often go undetected due to unavailability of required facilities and financial constraints in emerging economies like India. Only 1 of our VEOIBD patients was tested for receptor mutations.
The rate of steroid and biologic usage did not differ between the VEOIBD and LO-PIBD group. Although the disease severity was high and complicated and extensive disease phenotypes were common in our study, biologic usage was limited in our study. The risk of opportunistic infections, high prevalence of latent TB and of course the high cost with limited or no insurance coverage precludes biological therapy in the resource limited country settings like India [47]. Exclusive enteral nutrition was not used due to poor acceptability and compliance in addition to cost concerns. However, PEN was used as an adjunct to other therapies. PEN in CD is known to be associated with an extended period of disease remission [48].
We acknowledge certain limitations including, the relatively small number of children in VEOIBD group and inability to perform mutation analysis in a large fraction of children. We did not assess all the PIBD patients routinely for growth failure- so we could not assess the extent of growth failure in various groups which is important aspect of PIBD specially in CD [49]. It was also a single tertiary care center analysis with the possibility of overrepresentation of severe disease. Comparisons with the Western and other Asian data are at best tentative.
This is the first PIBD cohort from India highlighting the differences between VEOIBD and LO-PIBD in children. VEOIBD though uncommon was associated with more severe disease and poorer outcomes compared to later onset disease. VEOIBD in India was phenotypically similar to the West although later onset (6–17 years) disease phenotype differed. Large multicenter studies based on prospective databases are required to confirm whether PIBD in India is phenotypically different. We found that diagnostic delays were common and most children received multiple courses of antibiotics before confirmation of diagnosis. This would have significant long term prognostic implications. This study highlights the urgent need for increasing the awareness on the prevalence of IBD in the pediatric population amongst primary care physicians to ensure early diagnosis and timely treatment of this chronic disease.
Notes
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Author Contribution
Conceptualization: Banerjee R. Data curation: Pal P, Shava U, Ganesh G. Formal analysis: Pal P. Investigation: Pal P. Methodology: Banerjee R. Project administration: Banerjee R, Reddy DN. Resources: Banerjee R. Supervision: Banerjee R. Visualization: Banerjee R, Pal P. Writing - original draft: Banerjee R, Pal P, Nabi Z, Shava U, Ganesh G. Writing - review & editing: all authors. Approval of final manuscript: all authors.
Supplementary Material
Supplementary materials are available at the Intestinal Research website (https://www.irjournal.org).
Supplementary Table 1. Logistic Regression Analysis of Risk Factors of Surgery
ir-2020-00107-suppl1.pdfSupplementary Table 2. Comparison of Pediatric Inflammatory Bowel Disease Data with French Population-Based Registry (EPIMAD)
ir-2020-00107-suppl2.pdfSupplementary Table 3. PIBD Data Comparison with Indian, Asian and Western Studies
ir-2020-00107-suppl3.pdfSupplementary Table 4. Comparison of Adult and Pediatric Cohort in The Inflammatory Bowel Disease Registry
ir-2020-00107-suppl4.pdf