 |
 |
- Search
| Intest Res > Volume 17(3); 2019 > Article |
|
Abstract
Background/Aims
Methods
Results
NOTES
FINANCIAL SUPPORT
This work was supported by the Deutsche Forschungsgemeinschaft, Germany (Heisenberg Program).
CONFLICT OF INTEREST
J.W. received speakers honoray and/or consultant fees from Takeda, AbbVie, MSD, Ferring, Roche, Novartis, Janssen. T.K. received speakers honoray and/or consultant fees from Takeda, AbbVie, MSD, Novartis, Janssen. M.G. received speakers honoray and/or consultant fees from Takeda, AbbVie, MSD, Novartis, Janssen. Except for that, no potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION
Conceptualization: Saman S, Klag T, Wehkamp J. Methodology: Saman S, Klag T, Wehkamp J. Formal analysis: Saman S, Klag T, Wehkamp J, Wendler J, Goetz M. Funding acquisition: Wehkamp J, Malek NP. Project administration: Saman S, Klag T. Visualization: Saman S, Klag T, Wehkamp J, Wendler J, Malek NP. Writing - original draft: Saman S, Klag T, Wehkamp J. Writing - review and editing: all. Approval of final manuscript: all authors.
Fig. 1.
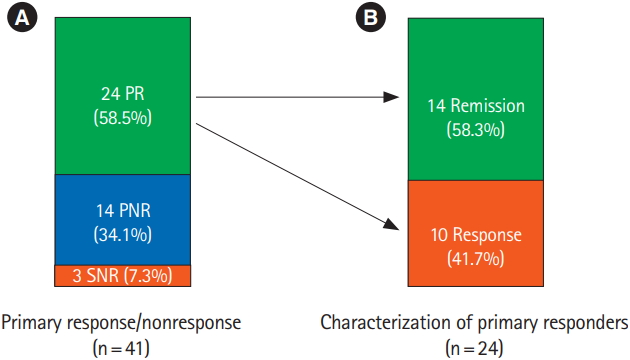
Fig. 2.
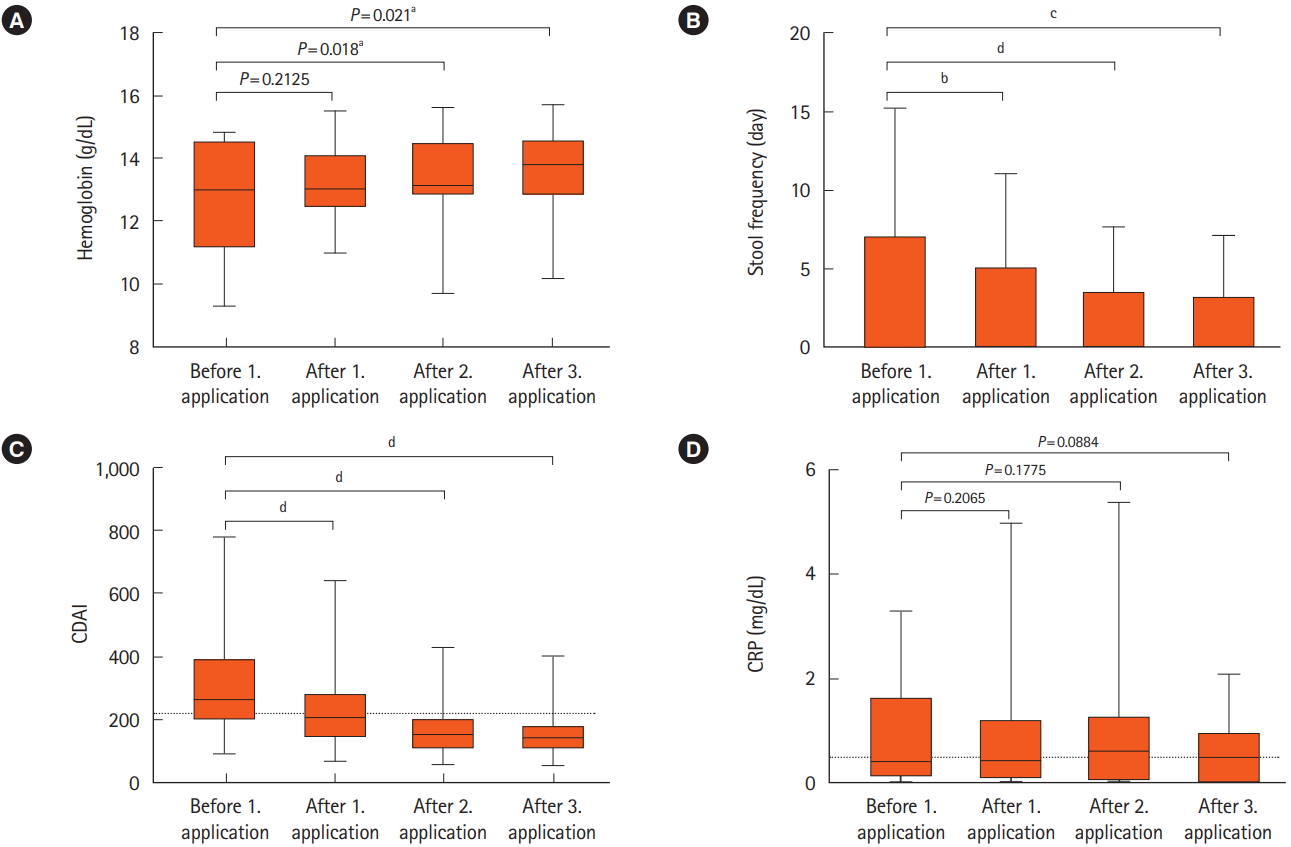
Fig. 3.
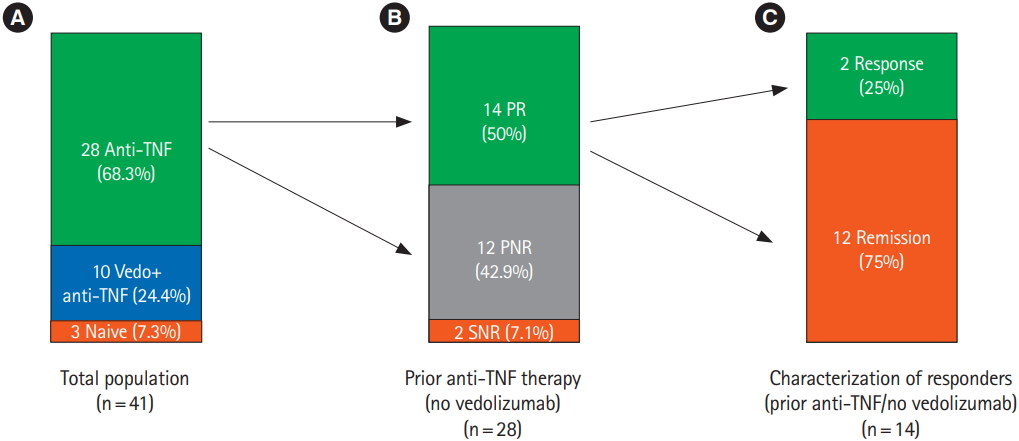
Fig. 4.

Fig. 5.

Table 1.
| Characteristic | Value (n = 13) |
|---|---|
| Sex | |
| Male | 10 (77) |
| Female | 3 (23) |
| Mean age (yr) | 36.3 |
| Median weight (kg) | 64.0 |
| Mean duration of disease (yr) | 11.1 |
| Median CRP (mg/dL) (n = 11)a | 0.8 |
| CRP level at baselinea | |
| ≤ 0.5 mg/dL | 4 (31) |
| > 0.5 mg/dL | 7 (54) |
| CDAI at baseline | |
| ≤ 150 | 3 (23) |
| 151-220 | 10 (77) |
| CD drugs at baseline | |
| Aminosalicylate drug | 2 (15) |
| Glucocorticoid | 5 (38) |
| Failure of previous treatment | |
| Any immunomodulatory drugs | 11 (85) |
| Anti-TNFs at baseline | |
| 1 | 10 (77) |
| >1 | 1 (7) |
| Anti-TNF+vedolizumab at baseline | 2 (75) |
| Failure criteria met | |
| Primary nonresponse | 5 (38) |
| Secondary nonresponse within 8 months | 0 |
| Unacceptable side effects | 1 (7) |
| Stoma | 3 (23) |
Table 2.
| Characteristic | Value (n = 28) |
|---|---|
| Sex | |
| Male | 9 (32) |
| Female | 19 (68) |
| Mean age (yr) | 35.5 |
| Median weight (kg) | 65.0 |
| Mean duration of disease (yr) | 14.3 |
| Median CRP (mg/dL) (n = 24)a | 1.3 |
| CRP level at baselinea | |
| ≤ 0.5 mg/dL | 9 (32) |
| > 0.5 mg/dL | 15 (53) |
| CDAI at baseline | |
| 221-450 | 25 (89) |
| > 450 | 3 (11) |
| CD drugs at baseline | |
| Aminosalicylate drug | 5 (18) |
| Glucocorticoid | 10 (36) |
| Failure of previous treatment | |
| Any immunomodulatory drugs | 27 (96) |
| Anti-TNFs at baseline | |
| 1 | 11 (39) |
| >1 | 9 (32) |
| Anti-TNF+vedolizumab at baseline | 8 (29) |
| Biologicals naive | 3 (11) |
| Failure criteria met | |
| Primary nonresponse | 9 (32) |
| Secondary nonresponse within 8 months | 3 (11) |
| Unacceptable side effects | 1 (3) |
| Stoma | 1 (3) |
Table 3.
| Normal group (CRP ≤ 0.5 mg/dL, n = 13) | Elevated group (CRP > 0.5 mg/dL, n = 22) | |
|---|---|---|
| Primary response | 9 (69) | 9 (41) |
| Primary nonresponse | 3 (23) | 11 (50) |
| Secondary nonresponse | 1 (8) | 2 (9) |
REFERENCES
- TOOLS








