Clinical features and outcomes in spontaneous intramural small bowel hematoma: cohort study and literature review
Article information
Abstract
Background/Aims
Spontaneous intramural small bowel hematoma (SISBH) is an extremely rare complication of anticoagulant or antiplatelet therapy. We assessed the clinical characteristics and outcomes of patients with SISBH according to the anatomical location of the hematoma.
Methods
From January 2003 to February 2016, medical records for all patients hospitalized for SISBH at 2 tertiary referral hospitals were retrospectively reviewed. The primary outcome was requirement for surgery.
Results
A total of 37 patients were enrolled. The mean age was 74.1 years. Among them, 33 patients (89.2%) were taking anticoagulant and/or antiplatelet agents. Duodenal intramural hematoma was detected in 4 patients (10.8%), jejunal in 16 (43.2%), and ileal in 17 (45.9%). Compared to jejunal and ileal involvement, duodenal intramural hematoma was significantly associated with high Charlson comorbidity index and low levels of white blood cells, hemoglobin, and platelets in the blood. SISBH in the duodenum was related to thrombocytopenia in 3 patients following systemic chemotherapy for malignancy. All patients with SISBH showed clinical improvement with conservative therapy. Mean length of hospital stay was 9.35 days. Independent predictors of a hospital stay of more than 7 days were body weight less than 60 kg (odds ratio [OR], 12.213; 95% confidence interval [CI], 1.755–84.998; P=0.011) and a history of cerebrovascular accidents (OR, 6.667; 95% CI, 1.121–39.650; P=0.037).
Conclusions
Compared to jejunal and ileal involvement, thrombocytopenia may result in spontaneous duodenal intramural hematoma among patients who are treated with systemic chemotherapy for malignancies. Patients with SISBH have excellent clinical outcomes with conservative therapy regardless of the anatomical location of the hematoma.
INTRODUCTION
Spontaneous intramural small bowel hematoma (SISBH) was initially described in 1838 [1], and has been often reported since the 1960s [2-4]. It is an extremely rare disease, with a reported incidence of 1 case per 2,500 patients receiving anticoagulant therapy per year in a retrospective epidemiologic survey [5]. The jejunum is the most commonly involved part of the small intestine followed by the ileum and duodenum [6,7]. The etiologic and clinical characteristics of SISBH based on the location of SISBH still remains unclear.
Anticoagulant or antiplatelet therapy is a risk factor for SISBH. Warfarin use was associated with most cases of SISBH [2-4,6-28] and heparin, low molecular weight heparin (LMWH), and aspirin can also induce SISBH [8,29-33]. The complications of SISBH such as intestinal obstruction, intestinal infarction, and pancreatitis with obstructive jaundice may require surgical interventions for clinical improvement [6,8,10,14,18,27-31,34]. However, the clinical prognosis of SISBH has not been fully determined due to its extremely low incidence. The aim of this study was to assess the clinical characteristics and outcomes of patients with SISBH according to the anatomical location of the hematoma.
METHODS
1. Patient Selection
From January 2003 to February 2016, the medical records of all consecutive patients who were hospitalized for SISBH at 2 tertiary referral hospitals in Korea (Seoul National University Hospital and Seoul National University Bundang Hospital) were retrospectively reviewed. All patients in whom intramural small intestinal hematoma was detected on the CT scan of the abdomen and pelvis were included in this study. Intramural small bowel hematoma was characterized as circumferential bowel wall thickening, luminal narrowing, and intestinal tract obstruction on the CT scan [6]. Any additional modality for small bowel evaluation including CT enterography, magnetic resonance enterography, capsule endoscopy and enteroscopy was not performed in the study population. Among these patients, those with a history of trauma or iatrogenic interventions including intraabdominal surgery or non-surgical procedures within 4 weeks from the presentation of the intramural hematoma were excluded. This study was approved by the Institutional Review Board Committee at the participating medical centers (SNUH IRB No. 1606-079-771; SNUBH IRB No. B-1608-359-401). The informed consent has been omitted. Because this study was a retrospective review of the patient’s medical records, there was no additional risk to the patient.
2. Study Protocol
Data such as age, sex, body weight, and medical history including diabetes mellitus, hypertension, cardiac arrhythmia, congestive heart failure, coronary heart disease, pulmonary thromboembolism, deep vein thrombosis, peripheral arterial disease, history of heart valve replacement, history of cerebrovascular accidents, and malignancy were collected. The Charlson comorbidity index was calculated to evaluate the severity of comorbidities in the study population. With each elevated level of the Charlson comorbidity index, there are stepwise increases in the cumulative mortality attributable to comorbid diseases [35]. The use of probable offending drugs including anticoagulants (warfarin, heparin, and novel oral anticoagulants such as dabigatran, rivaroxaban, and apixaban) and antiplatelets (aspirin, clopidogrel, ticlopidine, ticagrelor, and cilostazol) were reviewed. The GI symptoms associated with the presentation of SISBH and initial laboratory findings including white blood cell (WBC) count, hemoglobin level, platelet count, PT, aPTT, serum BUN, creatinine, albumin, and high-sensitivity CRP (hs-CRP) at the time of presentation were also collected. The primary outcome was the requirement for surgery. The secondary outcomes were lengths of fasting and hospital stay, and mortality rate. Oral feeding or enteral nutrition was considered according to the physician’s discretion in patients who were willing to try enteral nutrition and who had no obstructive GI symptoms including nausea, vomiting, and abdominal distension.
3. Statistical Analysis
Baseline characteristics and clinical outcomes according to the location of the hematoma in the small intestine were compared. Continuous variables were compared among the groups using one-way ANOVA with Bonferroni correction. Pearson chi-square test or Fisher exact test was used to calculate the statistical significance of the categorical variables. Continuous variables between prolonged (more than 7 days) and short (7 days or less) hospital stays were also compared using Student t-test. Variables considered predictive factors for prolonged hospital stay were assessed using univariate logistic regression analysis. In addition, variables that were significant in the univariate test were entered into the multivariate logistic regression model to identify independent predictive factors for prolonged hospital stay. P-values less than 0.05 were considered to be statistically significant. All statistical analyses were performed using the SPSS version 21.0 (IBM Corp., Armonk, NY, USA).
RESULTS
1. Baseline Characteristics of the Study Population
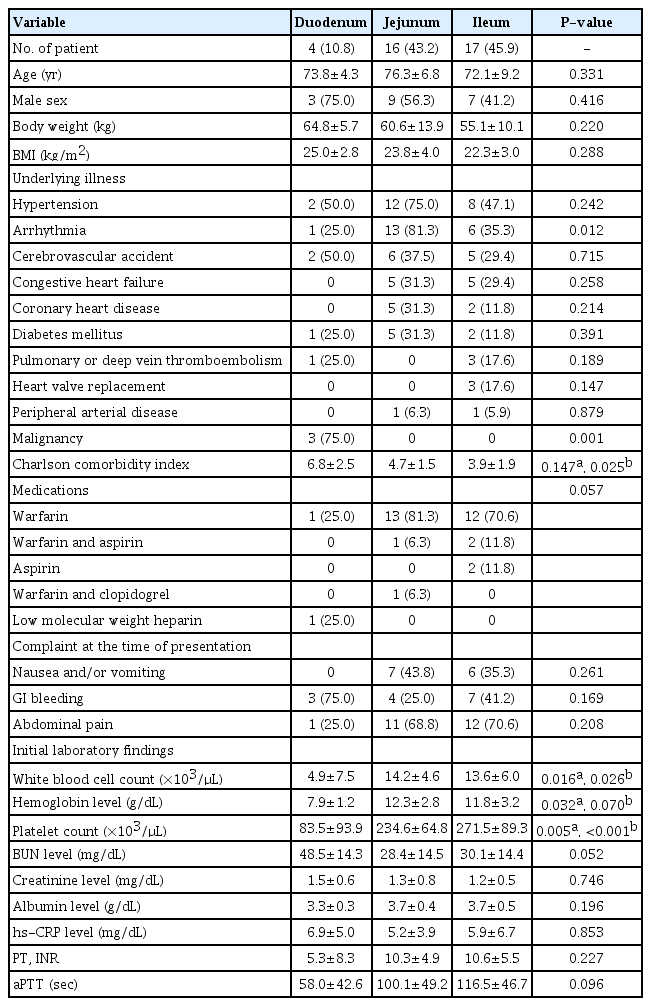
From January 2003 to February 2016, a total of 110,698 patients were treated with anticoagulant and/or antiplatelet therapy at the participating medical centers. Among them, a total of 37 consecutive patients with SISBH were enrolled in this study. The annual incidence of SISBH was estimated at 0.003% per year. The study population comprised 19 males (51.4%), and the mean age at presentation with SISBH was 74.1 years (range, 51–89 years). Among these patients, 35 (94.6%) had cardiovascular comorbidities including 22 with hypertension (59.5%), 20 with cardiac arrhythmia (54.1%), 13 with a history of cerebrovascular accidents (35.1%), 10 with congestive heart failure (27.0%), 7 with coronary heart diseases (18.9%), 4 with pulmonary embolism and/or deep vein thrombosis (10.8%), 3 with a history of heart valve replacement (8.1%), and 2 with peripheral arterial diseases (5.4%). Three patients (8.1%) had malignant tumors at the time that SISBH presented, all of whom were being treated with systemic chemotherapy. In total, 29 patients (78.4%) had 2 or more comorbidities, and the mean Charlson comorbidity score was 4.6 points (range, 2–10 points). Warfarin, aspirin, clopidogrel, and LMWH was taken by 30 (81.1%), 5 (13.5%), 1 (2.7%), and 1 (2.7%) patient, respectively. None of the study participants received novel oral anticoagulants. Only 1 patient without any underlying illness, who did not use any anticoagulant or antiplatelet therapies, showed prolongation of PT and aPTT during use of an herbal medication at the time of SISBH presentation. Abdominal pain was the most common complaint at the time of presentation (24 of 37 patients; 64.9%). Nausea and/or vomiting were present in 13 patients (35.1%) and GI bleeding developed in 14 (37.8%). Mean WBC count was 12.9×103/μL (range, 0.2–31.1×103/μL), mean hemoglobin level was 11.6 g/dL (range, 4.3–18.3 g/dL), and mean platelet count was 235×103/μL (range, 19–458×103/μL). Mean PT was 9.9 INR (range, 0.9–18.2 INR) and mean aPTT was 103 seconds (range, 22.7–180 seconds) (Table 1).
2. Clinical Characteristics of Patients with Spontaneous Intramural Hematoma According to Anatomical Location
Duodenal intramural hematoma was detected in 4 patients (10.8%), jejunal intramural hematoma in 16 (43.2%), and ileal intramural hematoma in 17 (45.9%). Cardiac arrhythmia occurred significantly more frequently in patients with jejunal intramural hematoma (81.3%) than in those with ileal (35.3%) and duodenal intramural (25.0%) hematomas (P =0.012). All 3 patients who were treated with systemic chemotherapy for malignancy had duodenal involvement. The Charlson comorbidity index in patients with duodenal intramural hematoma (6.8±2.5) was significantly higher than in patients with ileal intramural hematoma (3.9±1.9) (P =0.025). Patients who used warfarin had a predominance of jejunoileal involvement, although the difference among the locations of SISBH was not statistically significant. Compared to jejunal and ileal involvement, duodenal intramural hematoma was significantly associated with low levels of WBC, hemoglobin, and platelets in the blood (Table 1).
The clinical characteristics and treatment outcomes of the 4 patients with SISBH with duodenal involvement are shown in Supplementary Table 1. Among them, only 1 patient (25.0%; No. 4) had duodenal intramural hematoma related to coagulopathy, with a prolongation of PT and aPTT because of warfarin use. The 3 remaining patients with malignancy (No. 1-3) had duodenal intramural hematoma that developed in relation to thrombocytopenia within the normal range of PT and aPTT. Cytarabine and idarubicin had been administrated for the treatment of acute myeloid leukemia in 2 patients (patients No. 1 and 2) before presenting with a duodenal intramural hematoma. Patient No. 1 presented with sudden onset abdominal pain on the ninth day of the induction chemotherapy. At presentation, the patient’s platelet count was 66×103/μL, and the low platelet count persisted for about 4 weeks thereafter. Patient No. 2 presented with vomiting and hematemesis on the 62nd day of the induction chemotherapy. This patient’s initial platelet count was 28×103/μL. On the 5th day after admission, he was discharged without any remaining clinical signs or symptoms. Neither patient used any anticoagulant or antiplatelet therapies. Patient No. 3 had pancreatic cancer, which had been treated with gemcitabine and cisplatin, and had been on maintenance LMWH for 8 months to treat pulmonary thromboembolism. He visited the emergency department for vomiting with melena the 34th day after his last administration of chemotherapeutic agents. His initial platelet count was 19×103/μL. His symptoms subsided after discontinuing LMWH and correction of severe thrombocytopenia.
3. Primary and Secondary Clinical Outcomes
All patients were treated successfully with conservative therapy including fasting, blood component transfusion, and/or vitamin K injections. None of the patients underwent surgical therapy for SISBH-related complications. Overt GI bleeding was detected in 14 (37.8%) patients with SISBH. Among them, GI bleeding in all patients improved spontaneously by conservative therapy, except 1 patient who underwent angiographic embolization for ileal angiodysplasias although there was no evidence of active bleeding at the time of angiography. Multi-segmental hematoma in proximal to mid-jejunum was detected only in 1 patient (2.7%) with SISBH, an 80-year-old woman who had been taking warfarin for a cerebral infarction and cardiovascular disease. She complained of abdominal pain and nausea, and the initial laboratory findings showed prolonged PT (9.99 INR). She also improved without any complication by conservative therapy including fasting, parenteral nutrition, a transfusion of fresh frozen plasma and a vitamin K injection within 7 days of hospitalization. Moreover, there was no disease-related mortality in the study population. The mean length of fasting and the hospital stay were 3.38 days (range, 0–22 days) and 9.35 days (range, 1–70 days), respectively. Fourteen patients (37.8%) were hospitalized for more than 7 days (Table 2).
Among the 30 patients who were treated with warfarin, 26 (86.7%) continued warfarin after clinical improvement of the SISBH. Warfarin was switched to dual antiplatelet therapy with aspirin and clopidogrel in 2 patients, and to a novel oral anticoagulant (rivaroxaban and dabigatran) in 2 other patients. During the mean follow-up duration of 49.9 months (range, 5–153 months), recurrent SISBH developed in only 1 patient (2.7%) who was on warfarin therapy for a prosthetic heart valve 12 years after the presentation of the first SISBH.
4. Predictive Factors for a Longer Length of Hospital Stay
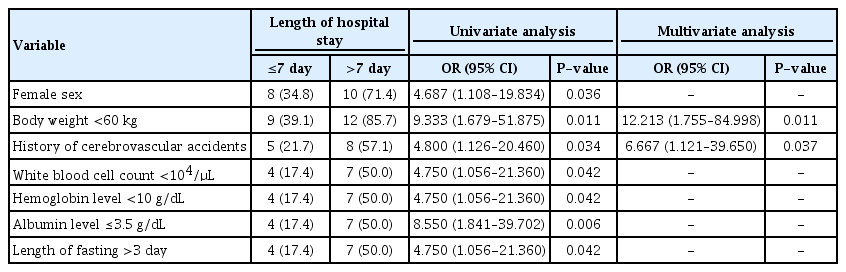
Prolonged hospital stay of more than 7 days was significantly associated with female gender (P =0.045), lower body weight (P =0.011), a history of cerebrovascular accidents (P =0.039), lower WBC counts (P =0.017), lower hemoglobin level (P =0.016), and lower albumin level (P =0.013), respectively (Supplementary Table 2). The multivariate analysis revealed that body weight less than 60 kg (OR, 12.213; 95% CI, 1.755–84.998; P =0.011) and a history of cerebrovascular accident (OR, 6.667; 95% CI, 1.121–39.650; P =0.037) were independent predictors for prolonged hospital stay (Table 3).
DISCUSSION
In this retrospective cohort study of 37 patients from 2 tertiary referral hospitals over 13 years, the clinical features and prognosis of SISBH were evaluated. To the best of our knowledge, this is the largest study done to determine the clinical characteristics of SISBH, especially according to the anatomical location of the hematoma.
A literature review of the MEDLINE database identified 40 published articles within the previous 30 years, with a total of 103 cases with SISBH that were analyzed [6-34,36-46]. The mean age of patients with SISBH was 62.2 years. Abdominal pain (72.8%) was the most frequent symptom at presentation in patients with SISBH, which is consistent with our results. Medication use (78.6%), including anticoagulant and antiplatelet therapy, was the most common cause of SISBH followed by hemophilia (4.9%), pancreatic diseases (3.9%), liver failure (1.9%), leukemia (1.0%), idiopathic thrombocytopenic purpura (1.0%), von Willebrand disease (1.0%), lupus (1.0%), and Glanzmann’s thrombasthenia (1.0%). Among the medications inducing SISBH, warfarin (73.8%) was the agent most frequently associated with the presentation of SISBH. In this study population, warfarin also contributed to the presentation of SISBH in 30 patients (81.1%). Moreover, more than 90% of patients with SISBH had cardiovascular comorbidities, of which hypertension and cardiac arrhythmia were the most common. These findings are consistent with a causal association between anticoagulant and antiplatelet therapy and SISBH, and suggest that cardiovascular diseases, not hemophilias, are the most critical comorbidities in patients with SISBH. This is because most studies included in the literature review were case reports or case series, which are descriptions of rare and unpredictable observations in clinical practice.
In the literature review, jejunal, ileal, and duodenal intramural hematoma was confirmed in 39 (37.9%), 25 (24.3%), and 18 (17.5%) patients, respectively. Three cases (2.9%) with SISBH involved diffuse segments of the small intestine and the involved segments of SISBH in the remaining 18 cases were unspecified. The predominance of jejunoileal involvement in SISBH was consistent with the results in our study. The reason why the duodenum is rarely involved in SISBH remains unclear. Because the duodenum is the shortest part of the small intestine, and is fixed and compressed by surrounding extraluminal organs, a duodenal intramural hematoma may be more easily compressed by the tissues and absorbed compared to those in the jejunum or ileum.
In this study, 2 patients with acute myeloid leukemia and thrombocytopenia experienced duodenal intramural hematomas. The other patient was diagnosed with duodenal intramural hematoma associated with newly detected thrombocytopenia while being treated with systemic chemotherapy for pancreatic cancer. The platelet counts in patients with duodenal intramural hematoma were significantly lower than those who had jejunoileal involvement. In addition, patients with SISBH with duodenal involvement had significantly more severe comorbidities as estimated by the Charlson comorbidity index compared to jejunoileal involvement. In the literature review, the proportion of cases where warfarin contributed to duodenal intramural hematoma was relatively small compared to the proportion with jejunoileal involvement. Also, case reports described that pancreatic diseases, including acute pancreatitis or pancreatic cancer, were related to duodenal intramural hematoma (Table 4) [34,42-44]. Taken together, the development of spontaneous duodenal intramural hematoma appears to be associated with thrombocytopenia in patients with severe comorbidities, such as malignancy or pancreatic diseases, rather than warfarin use. Duodenal involvement in SISBH is a rare event and its pathogenesis is difficult to clarify, but it seems to be caused by a different mechanism than jejunoileal intramural hematoma.
In the 103 patients with SISBH identified in the literature review, 18 (17.5%) required surgical treatment for peritonitis, intestinal obstruction, or necrotizing pancreatitis. There were 6 deaths (5.8%) and the causes of death were sepsis or multi-organ failure. In this study, however, all patients who presented with SISBH recovered after conservative management, without surgery. Recently, some reports showed that GI obstructive symptoms of SISBH could be treated conservatively without any sequelae, even in cases of extensively long segmental involvement of the hematoma [13,15,27,45,46]. Moreover, early detection of intramural hematoma is crucial because most patients with SISBH can be treated successfully without surgery [47]. Because the SISBH was detected and treated within 14 days (median, 2.5 days), the clinical prognosis of SISBH was favorable in our study.
In our study population, a prolonged hospital length of stay more than 7 days was significantly associated with low body weight of less than 60 kg and a history of cerebrovascular accident, respectively. Underweight is considered a risk for malnutrition [48], and was associated with a prolonged hospital stay in various populations such as patients undergoing pelvic exenteration or cardiac surgery [49,50]. Physical disabilities complicated by stroke were closely linked to a prolonged hospital length of stay, and poor participation in physical therapy of inpatients with functional impairments extended the hospital length of stay [51]. The acute symptoms caused by SISBH might delay the recovery of patients with functional impairment due to stroke.
In conclusion, compared to jejunal and ileal involvement, thrombocytopenia may result in spontaneous duodenal intramural hematoma among patients who are treated with systemic chemotherapy for malignancies. Patients with SISBH have excellent clinical outcomes with conservative therapy regardless of the anatomical location of the hematoma.
Notes
FINANCIAL SUPPORT
The authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION
Conceptualization: Kang EA and Han SJ. Methodology: Han SJ and Chun J. Data collecting and Formal analysis: Chun J, Lee HJ, Chung HS, and Yoon H. Project administration: Im JP, Kim SG, Kim N, and Lee DH. Visualization: Shin CM and Park YS. Writing-original draft: Han SJ and Chun J. Writingreview and editing: Kang EA, Chun J, Kim JS, and Jung HC. Approval of final manuscript: all authors.
Supplementary Materials
Supplementary Table 1.
Clinical Characteristics and Outcomes of 4 Patients with Duodenal Involvement
Supplementary Table 2.
Comparison of Characteristics and Outcomes Regarding Duration of Hospital Stay