INTRODUCTION
Crohn's disease (CD) is a chronic IBD that causes gastrointestinal symptoms such as abdominal pain, diarrhea, and bloody stool. CD can affect any part of the gastrointestinal tract, with the involvement of approximately 40% to 60% of the small intestine.1,2 The terminal ileum is the most common area affected by CD, which is accessible through colonoscopy.3 However, the proximal small bowel (SB) is affected in one third of patients with CD with normal ileocolonoscopy. Proximal SBCD can be difficult to diagnose because the proximal SB may not be accessible by conventional colonoscopy.
Chronic IBD is diagnosed by considering all the clinical, endoscopic, imaging, and histologic findings. Endoscopy plays an essential role in the diagnosis of IBD.4 Regarding the role of colonoscopy in the patients with CD, it is important to diagnose accurately, provide differential diagnosis of other inflammatory diseases, and evaluate the extent and activity of diseases.5 Furthermore, treatment response can be determined by evaluating the degree of mucosal healing and recurrence of inflammation.5 Strictures or bleeding complicated by chronic inflammation can be treated by balloon dilation or endoscopic hemostasis. The role of endoscopy is important for SBCD and colonic CD.
Over the past 10 years, SB examination achieved the advancement of radiologic imaging studies and endoscopic technologies. Traditional SB follow through (SBFT) or SB enteroclysis was recently replaced by CT enterography (CTE) or magnetic resonance enterography (MRE). In the endoscopic field, capsule endoscopy (CE) is a non-invasive luminal evaluation tool for the small intestine using a wireless capsule, which examines the entire SB. Balloon-assisted enteroscopy (BAE) is developed for endoscopic evaluation and therapeutic procedures of the small intestine.
These diagnostic and therapeutic technologies of SB diseases have their strengths and weaknesses. In this article, we reviewed the literature on the role of endoscopy in SBCD.
1. Capsule Endoscopy
CE was introduced in 2002 and revolutionized as small intestine imaging, which provides a noninvasive and sensitive method for complete visualization of the mucosal surface.6 Several CE commercial systems exist. They have different technologic features and software specifications: MiroCam, IntroMedic, Seoul, Korea; PillCam SB2, Given Imaging, Yoqneam, Israel; EndoCapsule, Olympus Europe GmbH, Hamburg, Germany; CapsoVision, Saratoga, CA, USA; and OMOM, Chongqing Jinshan Science, Beijing, China. The capsule is passively propelled by intestinal peristalsis while transferring captured images to an external receiver. CE is useful for detection of small mucosal lesions, which are not well detected in radiologic imaging studies.7 CE in patients with CD can identify CD-related mucosal change such as ulcers, erosions, erythema, aphthous lesions, or strictures.8
1) Patients With Suspected SBCD
Retrospective and prospective studies on CE showed that CE is a useful diagnostic method for the diagnosis of CD in patients with suspected SBCD who have negative colonoscopy results and radiologic imaging tests. A recent meta-analysis reported that diagnostic yields of CE in patients with suspected CD are significantly higher than that of SBFT, SB enteroclysis and CTE (CE, 52% vs. SBFT/SB enteroclysis, 16% and incremental yield, 32%; P<0.0001; 95% CI, 16%-48%; CE, 68% vs. CTE, 21% and incremental yield, 47%; P<0.00001; 95% CI, 31%-63%).7 Some studies have reported that CE is more useful than MRE for early CD or proximal SB lesion diagnosis.9 However, no statistical difference was found in the meta-analysis between CE and MRE for the diagnostic yields of suspected SBCD (CE, 55% vs. MRE, 45% and incremental yield, 10%; 95% CI, 14%-34%; P=0.43).7
These studies have limitations. The study results may have included false-positive patients because there are no definite criteria for the diagnosis of SBCD of CE, and criteria for suspected SBCD also vary from studies. Several studies showed that CE specificity is low, while its sensitivity is high (Table 1).9,10,11 However, negative predictive values of the CE findings are as high as 97% in patients with SBCD.12 The positive predictive values vary depending on the diagnostic criteria of SBCD in CE and the inclusion criteria of patients with suspected SBCD.12 Tukey et al. reported that the positive predictive value of CE was increased in patients selected with the International Conference on Capsule Endoscopy (ICCE) criteria and the presence of more than three ulcers in CE findings, whereas the positive predictive value was only 31% in patients with any criteria and any lesion among CE findings.12 Approximately 14% of healthy individuals have non-specific ulcers or erosions in CE,13 which could also appear in various SB diseases such as infection, ischemia, radiation injury, or NSAID-induced enteropathy.
The risk of capsule retention is low (1.6%) in patients suspected with SBCD without symptoms of obstruction, known stenosis, or history of SB resection.14,15,16 SB imaging or patency capsule is not routinely recommended before CE in patients with suspected SBCD without obstructive symptoms or known stenosis.14,15,16 Patency capsule or SB imaging such as CTE or MRE before CE is necessary for patients suspected with SBCD with symptoms of obstruction or suspected obstruction of abdominal imaging.17 Obtaining careful clinical history is the most important way to avoid the risk of capsule retention before CE.
2) Patients With Known SBCD
The examination for involvement of SB is necessary to determine the extent and location of inflammation in patients with established CD. Endoscopic investigation is informative in assessing remission maintenance, mucosal healing, or early inflammation recurrence. Furthermore, therapeutic endoscopic procedures could be needed for the diagnosis and treatment of CD complications. Endoscopic investigation with CE can be applied for patients with SBCD and colonic CD to assess mucosal healing, inflammation recurrence, or identify mucosal inflammation in patients with unexplained abdominal symptoms. The risk of capsule retention is 5% to 13% relatively higher in patients with established CD.14,15,18 SB cross-sectional imaging such as CTE or MRE is usually recommended before CE to identify strictures and assess the transmural and extraintestinal lesions of CD.17
Recent meta-analysis showed that CE has significantly higher diagnostic yields than SBFT or CTE in patients with known SBCD (CE, 71% vs. SBFT/SB enteroclysis, 36%; incremental yield, 38%; P<0.00001; 95% CI, 22%-54%; CE, 71% vs. CTE, 39%; incremental yield, 32%; P≤0.0001; 95% CI, 16%-47%).7 On the other hand, diagnostic yields of CE are similar with those of MRE (CE, 70% vs. MRE, 79%; incremental yield, -6%; P=0.65; 95% CI, -30%-19%).7 CTE or MRE is useful to identify the acute exacerbation of inflammation in SBCD.19 In cases of suspected active inflammation in SB imaging studies without inflammatory activity on colonoscopy, CE can detect inflammatory lesions of the small intestine and can affect therapeutic strategy.20,21 Moreover, CE can be helpful for the differential diagnosis between IBS and active CD inflammation, whether abdominal pain or diarrhea is from functional symptoms or active CD.
Mucosal healing is one of the most important end points of long-term prognosis related to the surgery risk or IBD complications.22 The remission of clinical symptoms was not well-correlated with endoscopic measures of disease activity. Deep remission, which is defined as endoscopic, biochemical and clinical remission, is an important treatment goal in CD to avoid hospitalization and the need for surgery. A recent study showed that SB inflammation is still detected in most of the patients with CD, who are in clinical and biomarker remission status. SB mucosal healing on CE is achieved in only 15.4% of patients in clinical remission.20 A retrospective study reported that CE findings have clinical impact on therapeutic plans in 52.3% of patients with established CD,21 whereas biomarkers such as CRP or fecal calprotectin are poorly correlated with significant SB inflammation. Therefore, CE findings in patients with established CD may help evaluate the mucosal healing status and response to therapy and can also affect the choice of therapeutic agents. Some studies assess the severity of SB inflammation by using the Lewis score or CECDAI, and further investigations are needed to evaluate these endoscopic scoring indicators.23,24
A diagnostic yield of CE has been compared with that of colonoscopy to assess relapse after surgical resection of CD. Some of the studies showed that the diagnostic yield of CE is higher than that of ileocolonoscopy (CE, 62% vs. ileocolonoscopy, 25%),25 whereas other studies showed that the diagnostic yield of CE is similar with that of ileocolonoscopy (CE, 62%-76% vs. ileocolonoscopy, 61%).26 CE is one of the less invasive options in evaluating postoperative recurrence of CD when colonoscopy is contraindicated or unsuccessful because the anastomosis is not readily accessible.
2. Balloon-assisted Enteroscopy
Two types of BAE are available widely. Double-balloon enteroscopy (DBE; Fujinon, Tokyo, Japan) uses two inflatable balloons at each distal end of the enteroscope and overtube, whereas single-balloon enteroscopy (Olympus, Tokyo, Japan) uses only one inflatable balloon at the distal end of overtube.27,28 Both BAE provide endoscopic examination of the entire small intestine through antegrade or retrograde approach, biopsies, and therapeutic endoscopic interventions such as balloon dilation or hemostasis.29
Endoscopic investigation more readily detects small mucosal lesions such as aphthae, erosions, or small ulcers than radiologic examinations. The diagnostic yields of BAE have been reported in 22%-70% of patients with suspected CD (Fig. 1).30,31,32,33 The complication rate of diagnostic BAE is approximately 1%, which is relatively low.30,32,33 A meta-analysis recently reported that the diagnostic yield of DBE for inflammatory lesions is approximately 16%, which has no significant difference compared with that of CE (18%).34 CE is relatively less invasive and provides information about the insertion route of the DBE. Therefore, CE is recommended as the first test before DBE in patients without fistula or stenosis.34
No sufficient data support BAE for the evaluation and management of SBCD. A recent study has compared the activity index of MRE and the simplified endoscopic activity score for CD of single-balloon enteroscopy in known patients with SBCD. This study showed that activity index of magnetic resonance well correlated with the simplified endoscopic activity score for CD in the SB.35 Furthermore, the study will be needed on whether radiologic imaging such as MRE or CTE would replace enteroscopic visualization to assess the inflammatory activity of SBCD.
BAE with SB biopsy is necessary for the diagnosis of CD or the exclusion of inflammatory diseases or neoplastic diseases in patients with no confirmative findings of CD in colonoscopy and suspected SBCD in the radiologic tests or CE.30,31,36,37,38,39 In addition, several recent studies showed that DBE detects SB lesions in patients with established CD (Table 2).40,41,42 Step-up therapy in 74% (26/35) of patients based on DBE findings led to clinical remission in 88% (23/26) of patients.40 BAE has an additional clinical impact on the decision of treatment options in patients with established CD with suspected SB activity and no distal disease activity.
A recent study reported that 22 patients with suspected SBCD and 43 patients with known SBCD underwent 78 BAEs, and 27.3% of patients with suspected CD had been diagnosed with CD, and 13.6%, with NSAID enteropathy.43 Among patients with known SBCD, active inflammation such as ulcers or stenosis was detected in 41.9% of patients, which led to a change in therapeutic plans.43 BAE has clinical impact on the diagnosis and treatment of patients with suspected or known SBCD.11
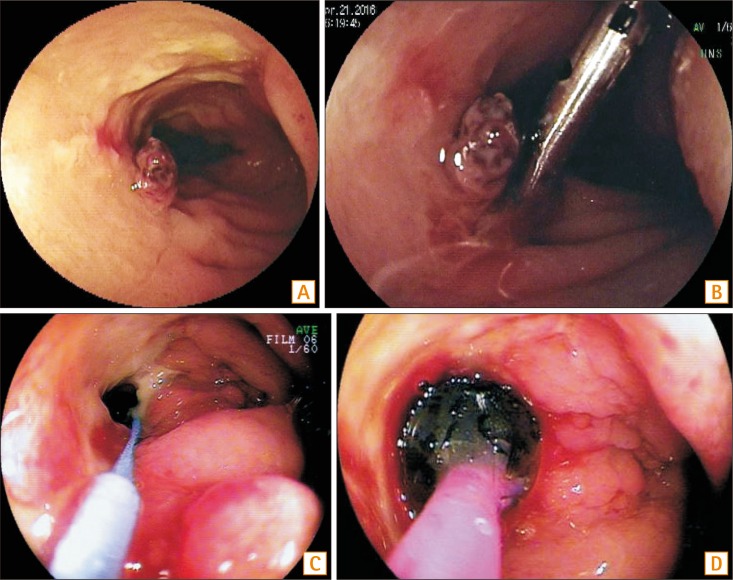
BAE provides therapeutic interventions such as hemostasis, balloon dilation, or retrieval of retained capsule in patients with suspected or known CD with bleeding, symptomatic strictures.31,43,44 Recent studies showed that technical success of balloon dilation is reported in approximately 60% to 80% of patients without deep ulcer or severe inflammation and with strictures less than 5 cm in length that are endoscopically accessible (Fig. 2).26,30,45 However, the risk of perforation following balloon dilation is reported to be 9%, which is higher than the complication rate of diagnostic BAE.30,31,34,40,45,46 Balloon dilation should be avoided in strictures longer than 5 cm or with fistula, severely inflamed, fixed and angulated, or deeply ulcerated stricture.
The advantages of BAE in patients with suspected CD or known CD include the endoscopic evaluation of atypical lesions, obtaining biopsies for histologic diagnosis, and therapeutic potentials such as balloon dilation or hemostasis. However, BAE is technically more complicated and invasive than CE or ileocolonoscopy. Adhesions limit the evaluation of the deeper part of the small intestine because of angulations and strictures, and there are higher risks of complications in patients with active stricturing CD. Therefore, BAE is recommended in patients with suspected CD when conventional radiographic studies or colonoscopy has shown inconclusive findings, and histologic evaluation is imperative for management. BAE is also indicated in patients with known CD when endoscopic visualization of the small intestine and biopsies are necessary to differentiate other diseases such as lymphoma, cancer, or tuberculosis, and when therapeutic procedures are essential to treat bleeding, strictures, or retained capsule.
CONCLUSIONS
SB is the only site involved in around 30% of patients with CD, although conventional ileocolonoscopy is the first diagnostic method in patients with suspected CD. Therefore, SB evaluation is an important part of the diagnosis of SBCD and continues to evolve because of technologic advances. SB endoscopy (CE or BAE) and cross-sectional imaging (CTE or MRE) have become necessary methods to diagnose and treat patients with CD. CE shows the highest diagnostic yield in patients with suspected CD, but CE has low positive predictive value and modest specificity and should be performed in patients without obstructive symptoms or known stenosis to avoid capsule retention. BAE has advantages in obtaining tissues to differentiate other diseases and therapeutic capabilities, including balloon dilation or hemostasis. On the other hand, CTE and MRE are noninvasive modalities that provide both luminal and extraluminal information. CTE or MRE should be used first in the presence of obstructive symptoms or known strictures. Growing efforts are nowadays made to develop several innovative SB imaging techniques. These imaging techniques can look beyond the conventional endoscopic techniques, thereby opening a new field to improve the diagnosis and treatment of SBCD, considering the pros and cons of each imaging technique.