 |
 |
- Search
| Intest Res > Volume 16(2); 2018 > Article |
|
Abstract
Background/Aims
Methods
Results
Conclusions
ACKNOWLEDGEMENTS
NOTES
FINANCIAL SUPPORT: These studies were sponsored by Pfizer Inc. Medical writing support under the guidance of the authors was provided by Daniel Binks PhD at CMC Connect, a division of Complete Medical Communications Ltd, Macclesfield, UK, and was funded by Pfizer Inc, New York, NY, USA in accordance with Good Publication Practice (GPP3) guidelines (Ann Intern Med 2015;163:461-464).
CONFLICT OF INTEREST: S.M. is supported for research financially by AbbVie GK, Janssen Pharma, and Pfizer Inc; and for lecture fees by Mitsubishi Tanabe Pharma Co., Ltd. M.W. received a research grant from Asahi Kasei Kuraray Medical Co., Ltd, Ajinomoto Pharma Co., Ltd, AbbVie GK, Eisai Co., Ltd, Kyorin Pharmaceutical Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, Otsuka Pharma Co., Ltd, Kyowa Hakko Kirin Co., Ltd, Zeria Pharmaceutical Co., Ltd, UCB Japan Co., Ltd, JIMRO Co., Ltd, Takeda Pharmaceutical Co., Ltd, Daiichi Sankyo Co., Ltd, Ono Pharmaceutical Co., Ltd, Gene Care Research Institute Co., Ltd, Astellas Pharma Inc., and MSD K.K.; and lecture fees from Mitsubishi Tanabe Pharma Co., Ltd, Eisai Co., Ltd, Ajinomoto Pharma Co., Ltd, and Takeda Pharmaceutical Co., Ltd. H.J.K., Y.H.K., and D.S.H. have no conflicts of interest to declare. T.H. has received grants and personal fees from Mitsubishi Tanabe Pharma Co., Ltd, EA Pharma Co., Ltd., JIMRO, and Zeria Pharma; grants from AbbVie; and personal fees from Takeda Pharma. H.Y., J.T., N.I., and S.A. are employees and stockholders of Pfizer Japan. I.K. was an employee of Pfizer Japan at the time these studies were conducted.
AUTHOR CONTRIBUTION: Investigation: S.M., M.W., H.J.K., Y.H.K., D.S.H., T.H. Conceptualization: H.Y., J.T., N.I., S.A., I.K. Formal analysis: N.I. Writing-original draft preparation: S.M., M.W., H.J.K., Y.H.K., D.S.H., H.Y., J.T., N.I., S.A., I.K., T.H. Writing-review & editing: S.M., M.W., H.J.K., Y.H.K., D.S.H., H.Y., J.T., N.I., S.A., I.K., T.H. Approval of final manuscript: all authors.
References
Fig. 1
Overview of the OCTAVE study design, including maintenance and open-label studies. Response in OCTAVE Induction 1 and 2 was defined as week 8 remission (total Mayo score ≤2, no individual subscore >1, and rectal bleeding subscore 0) or clinical response (decrease from baseline total Mayo score of ≥3 points and ≥30%, with an accompanying decrease in the rectal bleeding subscore of ≥1 or absolute rectal bleeding subscore ≤1). aFinal complete efficacy assessment at weeks 8 and 52; treatment continued to week 9 in OCTAVE Induction 1 and 2 and to week 53 in OCTAVE Sustain; bPatients in remission at open-label extension baseline received tofacitinib 5 mg BID; all other patients received tofacitinib 10 mg BID. BID, twice daily.
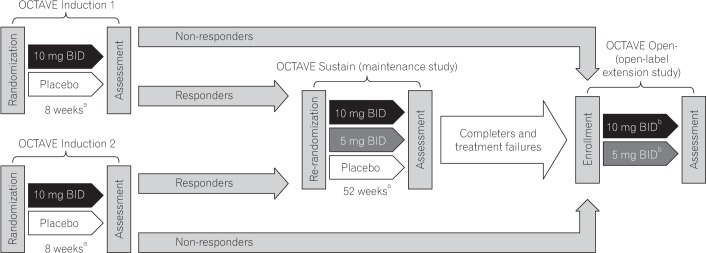
Fig. 2
Patient disposition of the East Asian population in (A) OCTAVE Induction 1 and 2 and (B) OCTAVE Sustain. Patients with insufficient clinical response included those who discontinued because of an adverse event (AE) of worsening UC. BID, twice daily.

Fig. 3
Proportion of East Asian patients in OCTAVE Induction 1 and 2 achieving (A) remission, (B) mucosal healing, (C) clinical response, (D) clinical remission, and (E) endoscopic remission at week 8 by treatment group, non-responder imputation. Remission: total Mayo score ≤2, no individual subscore >1, and rectal bleeding subscore 0; mucosal healing: Mayo endoscopic subscore 0 or 1; clinical response: decrease from baseline total Mayo score of ≥3 points and ≥30%, with an accompanying decrease in the rectal bleeding subscore of ≥1 or absolute rectal bleeding subscore ≤1; clinical remission: total Mayo score ≤2, no individual subscore >>1; endoscopic remission: Mayo endoscopic subscore 0. Data are from the full analysis set with non-responder imputation, central read. Δ, treatment effect size; BID, twice daily.
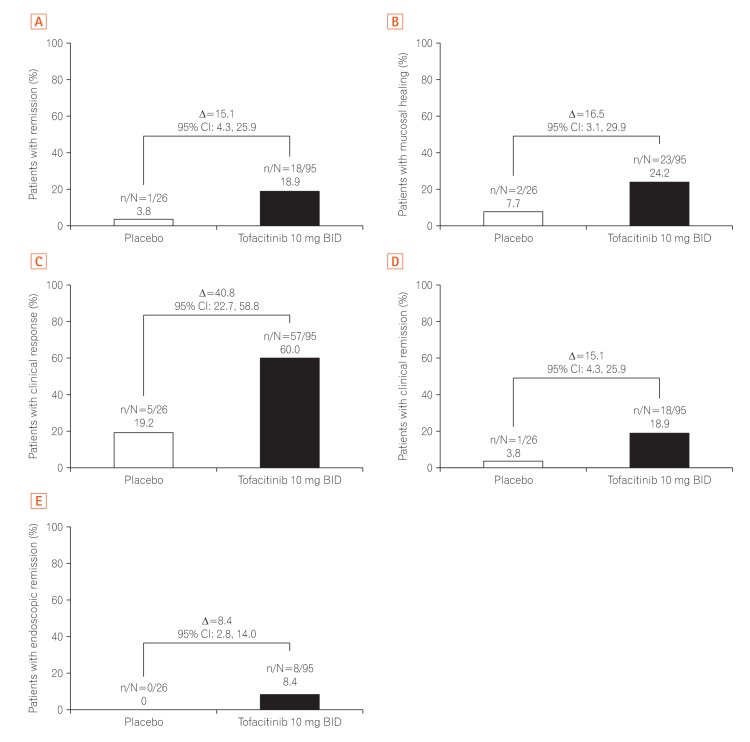
Fig. 4
Change from baseline in partial Mayo score over time in East Asian patients in (A) OCTAVE Induction 1 and 2 and (B) OCTAVE Sustain, by treatment group, observed case. Partial Mayo score: Mayo score excluding endoscopic findings subscore. Data are from the full analysis set, observed case based on descriptive statistics. BID, twice daily.
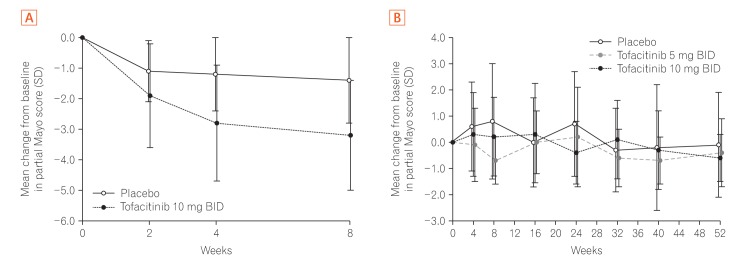
Fig. 5
Proportion of East Asian patients in OCTAVE Sustain achieving (A) remission, (B) mucosal healing, (C) sustained steroid-free remission among patients in remission at baseline, (D) clinical response, (E) clinical remission, and (F) endoscopic remission at week 52 by treatment group, non-responder imputation. Remission: total Mayo score ≤2, no individual subscore >1, and rectal bleeding subscore 0; mucosal healing: Mayo endoscopic subscore 0 or 1; sustained steroid-free remission: achieving remission at both weeks 24 and 52 and not requiring corticosteroids for ≥4 weeks before each visit; clinical response: decrease from the baseline total Mayo score of ≥3 points and ≥30%, with an accompanying decrease in the rectal bleeding subscore of ≥1 or absolute rectal bleeding subscore ≤1; clinical remission: total Mayo score ≤2, no individual subscore >1; endoscopic remission: Mayo endoscopic subscore 0. Data are from the full analysis set with non-responder imputation, central read. Δ, treatment effect size; BID, twice daily.

Table 1
Demographics and Baseline Disease Characteristics for the East Asian Population of Patients in OCTAVE Induction 1 and 2 and OCTAVE Sustain, by Treatment Group
Values are presented as number (%), mean±SD, or median (range). The baseline values for OCTAVE Sustain are at the time of randomization into OCTAVE Sustain, except where indicated.
aIndicated values are at baseline of OCTAVE Induction 1 and 2.
BID, twice daily; IBDQ, Inflammatory Bowel Disease Questionnaire; NA, not available; anti-TNF, anti-tumor necrosis factor.
Table 2
Summary of Safety in the East Asian Population of Patients in OCTAVE Induction 1 and 2 and OCTAVE Sustain
Values are presented as number (%). Patients with insufficient clinical response included those who discontinued because of an adverse event (AE) of worsening UC.
aThe 4 most frequently occurring AEs overall in OCTAVE Induction 1 and 2, and OCTAVE Sustain, are presented for each treatment group in OCTAVE Induction 1 and 2 and OCTAVE Sustain.
BID, twice daily; SAE, serious adverse event; RTI, respiratory tract infection.




 .
. .
.

