NUDT15 gene variants and thiopurine-induced leukopenia in patients with inflammatory bowel disease
Article information
Abstract
Thiopurine has been used to maintain remission and to reduce antidrug antibody formation in monoclonal antibody therapy in patients with inflammatory bowel disease (IBD). The use of thiopurine is limited by side effects such as leukopenia. Thiopurine S-methyltransferase (TPMT) variants are associated with thiopurine-induced leukopenia in Westerners, but the frequency of the risk alleles is low in Asians. Recently, a variant in the nudix hydrolase 15 (NUDT15) gene (R139C, c.415C > T) was reported to be associated with early severe leukopenia in Asians. NUDT15 is an enzyme that converts 6-thio-(deoxy)guanosine triphosphate (6-T(d)GTP) to 6-thio-(deoxy)guanosine monophosphate (6-T(d)GMTP). The R139C variant impairs the stability of the protein and increases incorporation of 6-TGTP and 6-TdGTP into RNA and DNA, respectively, resulting in leukopenia. The frequency of C/C, C/T, and T/T are approximately 80%, 20%, and 1%, respectively in East Asians. Early leukopenia occurred in less than 3% of patients with C/C and in around 20% of those with C/T, whereas it occurred in almost all patients with T/T. Patients homozygous for this variant also develop severe hair loss. The measurement of NUDT15 R139C can increase the safety of thiopurine dramatically and is a successful example of personalized medicine in the field of IBD.
INTRODUCTION
A purine analogue, 6-mercaptopurine (6-MP), initially developed as a treatment for acute leukemia [1], was found to have immunosuppressive properties [2]. Then, azathioprine (AZA), a prodrug of 6-MP, was first used to treat IBD in 1966 [3]. In patients with IBD, thiopurine has been used to maintain remission and, more recently, to reduce the production of antidrug antibodies in monoclonal antibody therapy. It is also used to treat other autoimmune diseases such as autoimmune hepatitis and systemic lupus erythematosus.
Despite the therapeutic efficacy of thiopurine in IBD, its use is limited because it may cause severe side effects such as leukopenia, which is more common in Asians than Westerners. The incidence of leukopenia is 15% to 40% in Asians [4-6], who seem to be more susceptible to thiopurine-induced leukopenia because the incidence of myelosuppression (including leukopenia, thrombocytopenia, and anemia) in Westerners is approximately 3% [7,8]. In particular, severe leukopenia occurs in approximately 1% of Asian patients early after starting thiopurine [9]. Therefore, while Western guidelines recommend the dose of AZA between 2 and 2.5 mg/kg [10], Asian guidelines recommend starting with a lower dose of 25 mg [11]. In Westerners, thiopurine S-methyltransferase (TPMT) gene variants (TPMT*2, *3A, *3B, *3C) are known to be associated with leukopenia. TPMT is one of the metabolizing enzymes of thiopurine. It is recommended that the dose of thiopurine be adjusted according to the TPMT gene variants [12]. In Asians, however, the allele frequency of the TPMT variants was only approximately 3% [13-16], and the TPMT variants alone do not predict leukopenia.
In 2014, a genome-wide association study in Korean IBD patients reported that a variant of the nudix (nucleoside diphosphate-linked moiety X)-type motif 15 (NUDT15) or nudix hydrolase 15 (official name) gene was associated with thiopurine-induced early leukopenia [16]. This variant (rs116855232) replaces arginine at position 139 of the protein with cysteine (R139C; p.Arg139Cys; c.415C>T). Almost simultaneously, a correlation between this NUDT15 gene variant and acute severe leukopenia caused by thiopurine was reported in Japanese patients with acute lymphocytic leukemia [17].
METABOLISM OF THIOPURINE
Orally administered AZA is absorbed from the gut and metabolized nonenzymatically to 6-MP in the body, which is then inactivated by both methylation and oxidation intracellularly (Fig. 1) [18]. TPMT is an enzyme that methylates and inactivates 6-MP to 6-methy-MP (6-MeMP). Xanthine oxidase (XO) metabolizes 6-MP to 6-thio-uric acid. 6-MP is eventually metabolized to 6-thio-guanine nucleotides (6-TGN). 6-TGN is a generic name for 6-thio-guanosine monophosphate (6-TGMP), 6-thio-guanosine diphosphate (6-TGDP), and 6-thio-guanosine triphosphate (6-TGTP) [19]. 6-TGDP is reduced to 6-thiodeoxyguanosine diphosphate (6-TdGDP), which is further phosphorylated to 6-thio-deoxyguanosine triphosphate (6-TdGTP). 6-TGTP is incorporated into RNA and 6-TdGTP into DNA, causing inhibition of RNA transcription and DNA replication, respectively, and leading to apoptosis of the cell. 6-TGTP also causes apoptosis of lymphocytes by inhibiting GTPase Rac1 [20]. In this metabolic pathway, reduced TPMT activity due to the genetic variants leads to increased 6-TGN levels and causes leukopenia.
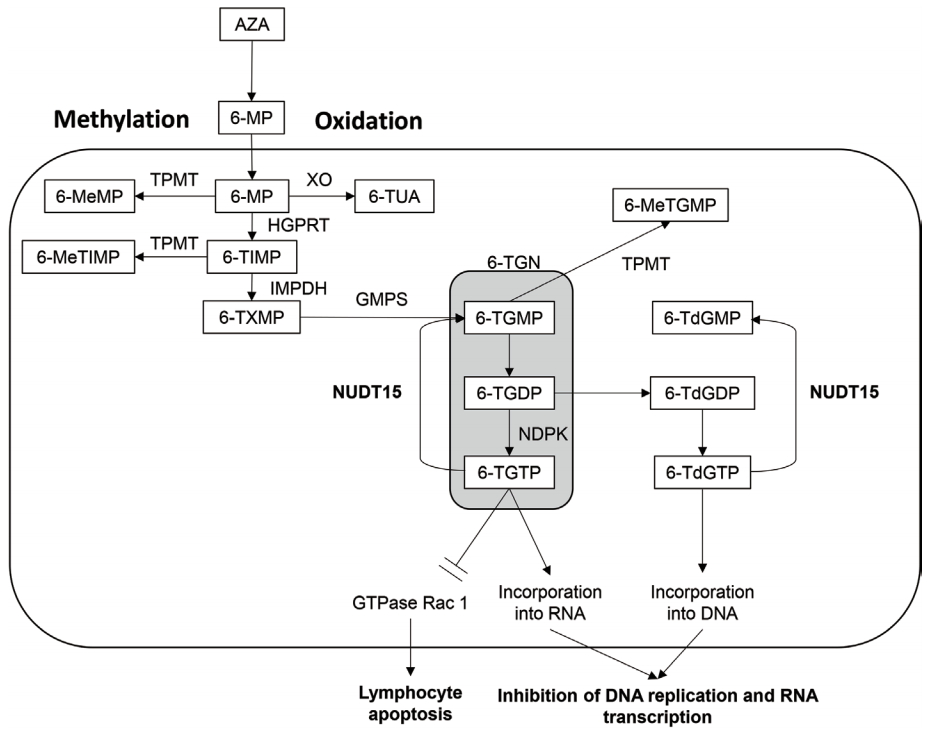
Metabolism of thiopurine [12,18,22,39]. 6-MeMP, 6-methyl-mercaptopurine; 6-MeTGMP, 6-methyl-thio-guanosine monophosphate; 6-MeTIMP, 6-methyl-thio-inosine monophosphate; 6-MP, 6-mercaptopurine; 6-TdGDP, 6-thio-deoxyguanosine diphosphate; 6-TdGMP, 6-thio-deoxyguanosine monophosphate; 6-TdGTP, 6-thio-deoxyguanosine triphosphate; 6-TGDP, 6-thio-guanosine diphosphate; 6-TGMP, 6-thio-guanosine monophosphate; 6-TGN, 6-thio-guanine nucleotides; 6-TGTP, 6-thio-guanosine triphosphate; 6-TIMP, 6-thioinosine monophosphate; 6-TXMP, 6-thio-xanthosine monophosphate; 6-TUA, 6-thio-uric acid; AZA, azathioprine; GMPS, guanosine monophosphate synthetase; HGPRT, hypoxanthine-guanine phosphoribosyltransferase; IMPDH, inosine monophosphate dehydrogenase; NDPK, nucleotide-diphosphate kinase; TPMT, thiopurine S-methyltransferase; XO, xanthine oxidase; NUDT15, nudix hydrolase 15.
ROLE OF NUDT15
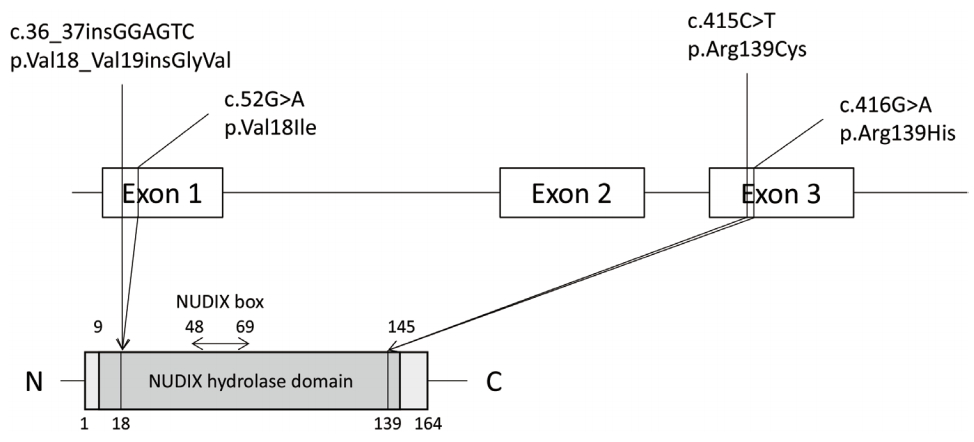
The function of NUDT15 was unknown when the correlation between its gene variants and thiopurine-induced leukopenia was reported. It was reported that the NUDT15 gene variant was not correlated with 6-TGN levels [9], suggesting that NUDT15 causes leukopenia independently of 6-TGN levels. Then, NUDT15 was found to be an enzyme that hydrolyzes 6-T(d) GTP to 6-T(d)GMP (Fig. 1) [21,22]. The NUDT15 gene is consisted of 3 exons and belongs to the NUDIX hydrolase family, which has the highly conserved NUDIX box and hydrolyzes nucleoside diphosphate liked to any moiety to nucleoside monophosphate (Fig. 2) [23]. Unlike the other NUDIX family proteins, the NUDT15 protein forms a homodimer [21].
The NUDT15 R139C variant impairs stabilization of its catalytic site and causes conformational change of the protein [22,24]. Thus, the variant in the NUDT15 gene reduces its enzymatic activity and increase the levels of 6-TGTP and 6-TdGTP. They are incorporated into RNA and DNA, respectively, causing leukopenia. These results can explain that the NUDT15 R139C gene variant does not correlate with 6-TGN levels because 6-TGN measures 6-TGMP, 6-TGDP, and 6-TGTP collectively. in vivo, administration of 6-MP to NUDT15 knockout mice increased the incorporation of 6-TdGTP into DNA [25]. In mice with the homologous mutation corresponding to the human NUDT15 R139C variant, a high dose of 6-MP (2 mg/kg) damages hematopoietic stem cells and progenitor cells and causes lethal leukopenia [26].
NUDT15 is an important enzyme in the metabolism of thiopurine, but its physiological function in vivo is still unknown. NUDT15 can hydrolyze 8-oxo-dGTP, one of the most common oxidative dNTP generated by oxidative stress and a potent mutagenic substrate for DNA synthesis, to 8-oxo-dGDP or 8-oxo-dGMP [27], but this effect of NUDT15 is of minor importance in vivo because depletion of NUDT15 has no effect on incorporation of 8-oxo-dGTP into DNA in vivo [21].
FREQUENCY OF NUDT15 GENE VARIANTS
Table 1 shows the frequency of the NUDT15 R139C variant in Asians; the frequencies of C/C, C/T, and T/T are approximately 80%, 20%, and 1%–5%, respectively [9,28-34]. It should be noted that most of the studies are retrospective and may overestimate the frequency of T/T. The only prospective study by Chang et al. [28] reported that the frequency of T/T is 1.2%.
The NUDT15 R139C variant is also found in South Americans with Native American ancestry [35]. However, in the Middle East, the frequency of this variant is less than one-tenth of East Asians [36]. The frequency of the NUDT15 R139C variant is also extremely low in Europeans and Africans [37]; however, the allele frequency of another variant of the NUDT15 gene, p.Gly17_Val18del, is observed at about 2% in Europeans. This variant was also reported to correlate with thiopurine-induced leukopenia [38].
To date, several variants have been reported in the NUDT15 gene, of which 4 variants (c.36_37insGGAGTC, c.52G > A, c.415C > T, c.416G > A) are mainly observed (Fig. 2). Six haplotypes combining these variants are reported (Table 2) [37]. The haplotypes carrying p.Arg139Cys (*2, *3) cause an extremely low enzymatic activity; the other variant haplotypes (*4, *5, *6) results in an intermediate decrease in enzymatic activity.37,39 Other rare variants include p.Arg34Thr, p.Lys35Glu, p.Gly17_Val18del, p.Met1Thr, p.Arg10Trp, and p.Gly47Arg [39]. The effects of these variants on thiopurine-induced leukopenia have rarely been tested in patients with IBD.
THIOPURINE-INDUCED EARLY SEVERE LEUKOPENIA AND NUDT15 GENE VARIANTS
Table 3 presents the association between the incidence of early leukopenia and NUDT15 R139C variant in studies in which early leukopenia was defined as a white bold cell count < 3,000 cells/mm3 within 8 weeks after initiation of thiopurine [9,16,29-31]. Early leukopenia occurred in less than 3% of patients with C/C and in around 20% of those with C/T, whereas it occurred in almost all patients with T/T. In addition to the R139C variant, a Chinese group reported that the c.36_37insGAGTC and c.52G > A variants were also associated with leukopenia and the combination of these variants can predict leukopenia more accurately [32]. The effects of variants other than R139C need to be further investigated.
A Korean prospective study confirmed the usefulness of measuring the NUDT15 R139C gene variant prior to thiopurine administration [28]. In this study, 182 patients with IBD were randomly assigned to receive or not receive genetic testing (NUDT15, FTO [fat mass and obesity-associated protein], TPMT) prior to azathioprine administration. Azathioprine was started at 50 mg and gradually increased to 2.0–2.5 mg/kg in patients with no genetic variant in the genotyping group and in those in the non-genotyping group. In patients homozygous for any risk variant, azathioprine was not recommended to be administered. The incidence of myelosuppression during the study period was significantly higher in the non-genotyping group than in the genotyping group (35.9% vs. 16.7%). The heterozygous NUDT15 variant was responsible for most of leukopenia.
Based on these accumulating evidence, the NUDT15 gene variant is described in the recent guideline as a test to be carried out before thiopurine administration [12]. In Japan, the measurement of NUDT15 R139C variant was approved for clinical use in February 2019.
Thiopurine is contraindicated in patients with T/T; the optimal way to administer thiopurine in patients with C/T remains undetermined. A large Japanese multicenter retrospective study showed that the time to leukopenia was shorter and tolerated doses of thiopurine were lower in patients with C/T or T/T than those with C/C [40]. Another study demonstrated that the decrease in leukocyte and platelet count persisted longer (up to 6 months) in patients heterozygous for this variant than those without this variant [41]. Careful monitoring of leukocyte counts over time is required in such patients.
OTHER SIDE EFFECTS OF THIOPURINES AND NUDT15 GENE VARIANTS
Thiopurine has also been associated with severe hair loss, a side effect not seen in Westerners, and unique to Asians. This severe hair loss is also associated with the NUDT15 R139C variant [30]. Severe hair loss is unavoidable when taking thiopurine in patients homozygous for this variant (the incident rate is 100%) [30]. It does not occur in patients without or heterozygous for the variant. Thus, severe thiopurine-induced hair loss completely depends on the homozygous NUDT15 gene variant. It was reported that gastrointestinal symptoms may also be associated with the NUDT15 variant [31]. Other side effects including liver damage, pancreatitis, fever, and skin symptoms do not appear to be associated with the variant [40].
CONCLUSIONS
Severe leukopenia and hair loss, the primary concerns in thiopurine administration in Asians, are almost completely predictable with the NUDT15 R139C gene variant. This discovery dramatically increased the safety of thiopurine. This represents an important successful example of personalized medicine in the field of IBD. Thiopurine, an old drug, is still a key remission-maintaining medication in IBD. Measuring the NUDT15 R139C variant, use of thiopurine is expected to be optimized. In contrast, other side effects of thiopurine including liver damage, pancreatitis, and lymphoma cannot be prevented by the NUDT15 variant. Further research is needed to reduce these risks associated with thiopurine.
Notes
FINANCIAL SUPPORT
The authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICT OF INTEREST
Matsuoka K received lecture fees from Mitsubishi Tanabe Pharma, Takeda Pharmaceutical, Janssen Pharmaceutical, Abbvie, EA Pharma, Pfizer, Mochida Pharmaceutical, Kyorin Pharmaceutical, Zeria Pharmaceutical, Kissei Pharmaceutical, Nippon Kayaku, Thermo Fisher Scientific, Alfresa Pharma, JIMRO, Miyarisan Pharmaceutical; consultancy fees from Thermo Fisher Scientific, Alfresa Pharma, EA Pharma, Takeda Pharmaceutical, Janssen, Abbvie, Mitsubishi Tanabe Pharma; research grants from Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, Abbvie, Takeda Pharmaceutical, Pfizer, Nippon Kayaku, Shionogi Pharmaceutical, EA Pharma, Zeria Pharmaceutical, Kissei Pharmaceutical.
AUTHOR CONTRIBUTION
Writing and approval of final manuscript: Matsuoka K.